The quest for interventions that can slow or reverse aspects of aging is one of the most ambitious goals in modern medicine. A fascinating area of this research focuses on developing “exercise mimetics”—compounds that replicate the health benefits of physical activity at a molecular level. One of the most talked-about experimental compounds in this category is SLU-PP-332. Despite being commonly referred to online as a ‘peptide,’ SLU-PP-332 is a synthetic small-molecule ERR agonist, not a peptide hormone or amino-acid–based compound. It is a small molecule designed to activate the same metabolic pathways switched on by endurance training.
This article provides a comprehensive, evidence-based overview of the SLU-PP-332 compound, its mechanism of action, the findings from preclinical studies, and its place within the broader landscape of exercise-mimetic compounds. It is crucial to state from the outset that SLU-PP-332 is an experimental compound that has only been studied in animal models. It is not approved for human use, and its safety and efficacy in humans are completely unknown.
The Science of Cellular Energy: Understanding Mitochondria and Aging
To understand the significance of a compound like SLU-PP-332, we must first appreciate the role of mitochondria. Often called the “powerhouses” of the cell, these tiny organelles are responsible for generating over 90% of the energy (in the form of adenosine triphosphate, or ATP) that our bodies need to function.
As we age, our mitochondria tend to become less efficient and more dysfunctional. This decline is considered one of the key hallmarks of the aging process. Dysfunctional mitochondria produce less energy and generate more harmful reactive oxygen species (ROS), leading to a vicious cycle of cellular damage. This age-related mitochondrial decline contributes to a wide range of issues, including muscle weakness (sarcopenia), reduced physical endurance, metabolic syndrome, and an increased risk of neurodegenerative diseases.
Consequently, strategies aimed at improving mitochondrial health—either by repairing existing mitochondria or by creating new ones (a process called mitochondrial biogenesis)—are a major focus of longevity and aging science. This is precisely where exercise mimetics like SLU-PP-332 come into play.
Mechanism of Action: How Does SLU-PP-332 Work?
The primary appeal of SLU-PP-332 lies in its unique and targeted mechanism of action. Unlike broader metabolic activators, it hones in on a specific family of proteins that are central to endurance and metabolism. The process can be broken down into a few key steps.
Targeting the ERRα, β, and γ Receptors
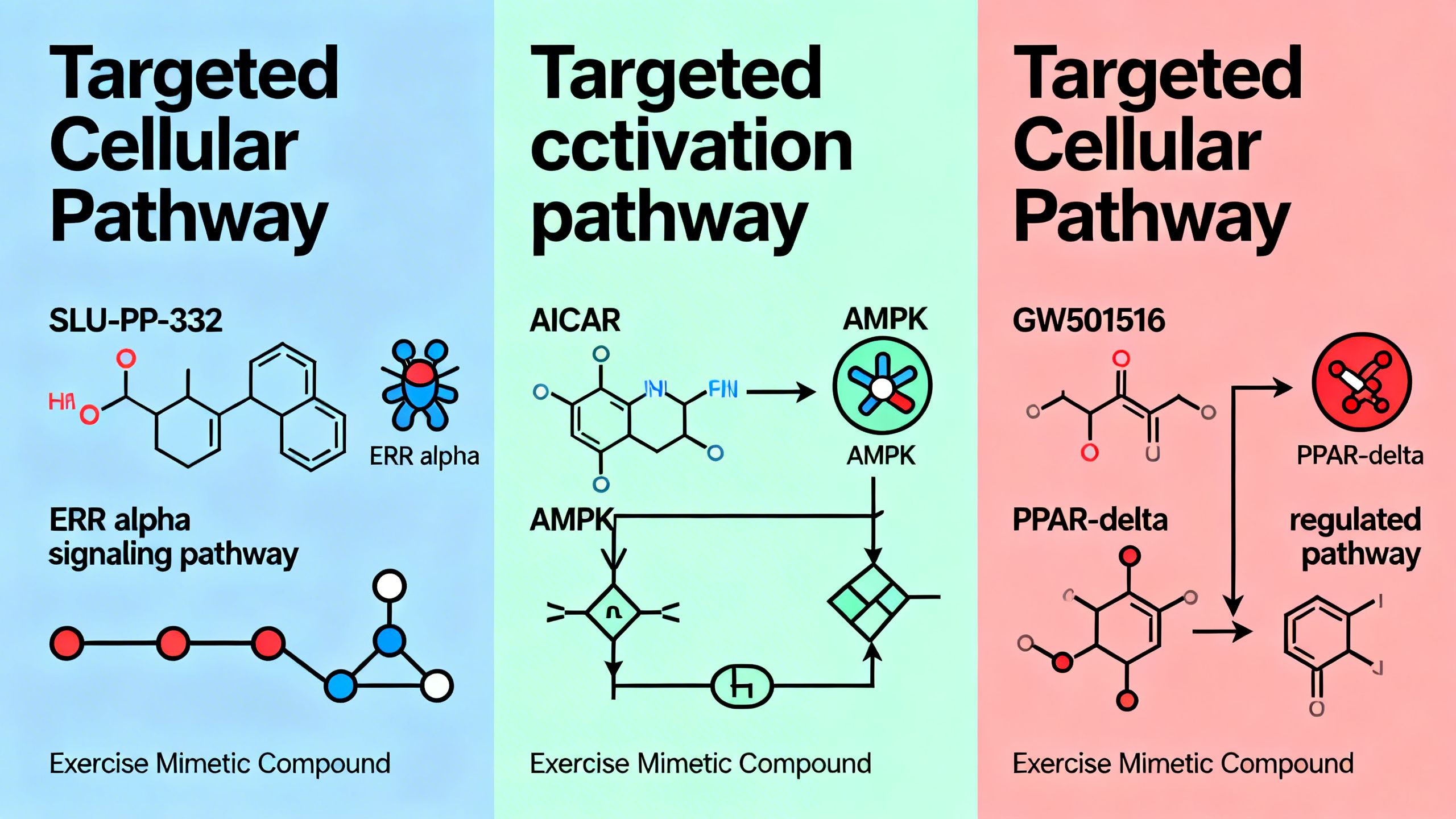
SLU-PP-332 is an agonist for the Estrogen-Related Receptors: ERRα, ERRβ, and ERRγ. Despite their name, these receptors do not bind to estrogen. They are classified as “orphan nuclear receptors” because their natural activating ligand (the molecule that turns them on) was initially unknown. These ERRs act as master genetic switches that control a vast network of genes involved in cellular energy metabolism.
- ERRα is highly expressed in tissues with high energy demands, such as the heart, skeletal muscle, and brown adipose tissue. It is a key regulator of fatty acid oxidation (burning fat for fuel) and mitochondrial biogenesis.
- ERRβ and ERRγ play complementary roles in development, pluripotency, and metabolic regulation in various tissues.
By activating all three types of ERR receptors, SLU-PP-332 initiates a coordinated, system-wide shift towards a more energy-efficient and oxidative metabolic state, much like the body’s response to consistent endurance training.
Activating the PGC-1α Pathway
The activation of ERRs by SLU-PP-332 doesn’t happen in a vacuum. These receptors work in close partnership with a coactivator protein called PGC-1α (Peroxisome proliferator-activated receptor-gamma coactivator 1-alpha). PGC-1α is often hailed as the “master regulator” of mitochondrial biogenesis.
When you engage in endurance exercise, PGC-1α is naturally activated in your muscles. It then works with transcription factors, including the ERRs, to turn on the genes needed to build new mitochondria, increase blood vessel formation, and enhance the muscle’s ability to use oxygen. SLU-PP-332 effectively hijacks this system, activating the ERRs, which then robustly recruit PGC-1α to initiate this same genetic program, leading to increased mitochondrial density and function, even in the absence of a physical training stimulus.
Simulating Endurance Signaling Without Exercise
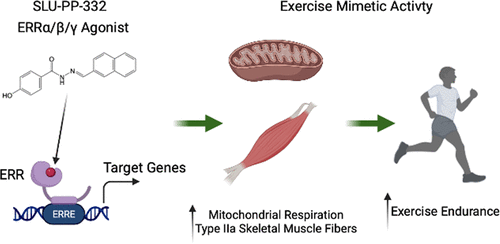
The ultimate effect of the ERR/PGC-1α activation by SLU-PP-332 is a profound change in muscle physiology. The compound encourages a shift in muscle fiber composition from the fast-twitch, glycolytic fibers (used for short bursts of power) to the slow-twitch, oxidative fibers. These slow-twitch fibers are rich in mitochondria, have a greater blood supply, and are highly resistant to fatigue, making them ideal for endurance activities.
This molecular reprogramming is the essence of the slu-pp-332 exercise mimetic effect. It tricks the muscle cells into believing they have been undergoing endurance training, prompting them to adapt accordingly by boosting their metabolic machinery.
To summarise, SLU‑PP‑332 binds and activates ERRα, ERRβ, and ERRγ → these nuclear receptors recruit the coactivator PGC‑1α → PGC‑1α turns on genes that build new mitochondria and improve muscle oxidative capacity → resulting in enhanced endurance and metabolic efficiency, even without physical exercise.
Evidence from Preclinical Research: What Animal Studies Show
It is essential to reiterate that all current evidence for SLU-PP-332’s effects comes from preclinical studies, primarily in mice. These findings are promising but cannot be assumed to translate directly to humans. The complexity of human physiology, metabolism, and potential for off-target effects means that what works in a mouse may not work, or may not be safe, in a person.
Enhanced Endurance and Physical Performance
The most cited slu-pp-332 peptide research involves its effects on physical endurance. In a key study, sedentary mice were treated with SLU-PP-332. When placed on a treadmill, these mice were able to run approximately 70% longer and farther than the untreated sedentary mice. Their performance became comparable to that of mice that had undergone a month-long endurance training regimen. This demonstrated that the compound could confer the benefits of exercise on physical stamina without the exercise itself.
Metabolic Benefits and Weight Management
Beyond pure performance, animal studies have highlighted significant metabolic benefits. Mice treated with SLU-PP-332 showed resistance to weight gain, even when fed a high-fat diet. This is attributed to an increase in their basal metabolic rate and energy expenditure. The compound effectively turned their metabolism up, causing them to burn more calories at rest.
Furthermore, the mice exhibited improved glucose tolerance and insulin sensitivity. This suggests that SLU-PP-332 could have potential applications in managing metabolic disorders like type 2 diabetes, a condition closely linked to mitochondrial dysfunction and a sedentary lifestyle.
Changes in Muscle Fiber Type
Analysis of the muscle tissue from treated mice confirmed the mechanism of action. Researchers observed a significant increase in the number of oxidative, slow-twitch muscle fibers. This physiological adaptation is a classic hallmark of endurance training and directly explains the observed improvements in stamina and metabolic health. The muscles were literally rebuilt to be more efficient at using fat for fuel and resisting fatigue.
SLU-PP-332 vs. Real Exercise: A Realistic Comparison
While the term “exercise in a pill” is catchy, it’s also a significant oversimplification. SLU-PP-332 and other exercise mimetic compounds aging research focuses on are not replacements for actual physical activity. Exercise confers a vast array of benefits that a single molecule is unlikely to ever replicate fully.
| Benefit | Physical Exercise | SLU-PP-332 (Based on Animal Data) |
|---|---|---|
| Mitochondrial Biogenesis | Yes | Yes |
| Improved Endurance | Yes | Yes |
| Improved Insulin Sensitivity | Yes | Yes |
| Cardiovascular Adaptation | Yes (stronger heart, lower resting HR) | Unknown/Unlikely |
| Increased Bone Density | Yes (with weight-bearing exercise) | No |
| Mental Health Benefits | Yes (endorphin release, reduced anxiety) | No |
| Improved Motor Skills/Coordination | Yes | No |
| Lymphatic System Stimulation | Yes | No |
Therefore, the most realistic therapeutic vision for SLU-PP-332 is not as a lifestyle shortcut for the healthy but as a potential treatment for individuals who are physically unable to exercise. This could include the elderly suffering from severe sarcopenia, patients with muscular dystrophy, individuals with spinal cord injuries, or those on prolonged bed rest. For these populations, a compound that could prevent muscle wasting and metabolic decline would be transformative.
The Landscape of Exercise Mimetics and Metabolic Modulators
SLU-PP-332 is part of a growing class of compounds targeting metabolic pathways related to exercise and aging. Understanding its context requires looking at other notable players.
AICAR (Acadesine)
AICAR is often considered a first-generation exercise mimetic. It works by activating AMPK, another master metabolic regulator that is also switched on by exercise. In animal studies, AICAR improved endurance and insulin sensitivity. However, its development has been hampered by very poor oral bioavailability and a short half-life, requiring large and frequent injections. Human trials have focused on its potential to protect the heart during surgery, not as a general exercise mimetic.
GW501516 (Cardarine)
Cardarine is a potent PPARδ agonist, a pathway related to the ERRs. It showed incredibly powerful endurance-enhancing effects in animal studies, far exceeding those of AICAR. However, its development was famously terminated after long-term animal studies revealed that it caused cancer to develop rapidly in multiple organs. This serves as a stark and critical warning about the potential dangers of chronically activating powerful metabolic pathways. The case of Cardarine underscores the absolute necessity of long-term safety studies for any new exercise mimetic.
Nicotinamide Riboside (NR) and Mononucleotide (NMN)
NR and NMN are not direct exercise mimetics but are precursors to the vital coenzyme NAD+. NAD+ levels decline with age, and this decline is linked to mitochondrial dysfunction. By boosting NAD+, these supplements aim to support mitochondrial function and the activity of sirtuins, another family of proteins involved in longevity. Human evidence is mixed but growing. Some studies show improvements in metabolic markers, while effects on physical performance and strength have been less consistent. They are generally considered safe but work more as a support nutrient than a powerful signaling molecule like SLU-PP-332.
Urolithin A
Urolithin A is a fascinating compound produced by our gut bacteria from ellagitannins found in foods like pomegranates, berries, and nuts. Its primary mechanism is stimulating “mitophagy”—the selective clearing and recycling of old, damaged mitochondria. Human clinical trials have shown that Urolithin A supplementation can improve mitochondrial gene expression and has led to modest improvements in muscle endurance in older adults. It represents a gentler, more restorative approach to slu-pp-332 mitochondrial function enhancement.
Safety, Toxicology, and the Unanswered Questions of SLU-PP-332 Peptide Research
The most significant section of this article is also the shortest, for one simple reason: there is no human safety data for SLU-PP-332. Its use is confined to laboratory research.
Any discussion of safety is therefore theoretical and based on its mechanism and data from other compounds:
- Unknown Long-Term Effects: The primary concern is the unknown consequence of chronically activating the ERR receptors. These are powerful master regulators, and long-term artificial stimulation could have unforeseen off-target effects or disrupt the body’s natural metabolic rhythms.
- Cancer Risk: The shadow of Cardarine (GW501516) looms large over this field of research. While SLU-PP-332 targets a different receptor (ERRs vs. PPARδ), they are both nuclear receptors that control cell growth and metabolism. The potential for increased risk of certain cancers with long-term use cannot be ruled out without extensive, multi-year toxicology studies.
- The Black Market Danger: Because of the hype surrounding it, SLU-PP-332 is illicitly sold online as a “research chemical.” Purchasing or using these products is exceptionally dangerous. There is no way to verify the identity, purity, or dosage of the substance. These products are often contaminated with solvents, heavy metals, or other dangerous compounds. Using an unverified, experimental substance with no human safety profile is a reckless gamble with one’s health.
The Future of SLU-PP-332 and Experimental Longevity Compounds
The path from a promising preclinical compound to an approved human therapeutic is long, arduous, and fraught with failure. For SLU-PP-332, the journey has only just begun.
The next steps in metabolic aging research peptides like this one would involve:
- Extensive Preclinical Toxicology: Comprehensive studies in multiple animal species to assess long-term safety and identify any potential for carcinogenicity or other adverse effects.
- Phase I Clinical Trials: If preclinical safety is established, the first human trials would begin. These involve giving small, carefully controlled doses to a small group of healthy volunteers to assess safety, tolerability, and how the drug is metabolized in the human body.
- Phase II Clinical Trials: The compound would then be tested in a larger group of patients with a specific condition (e.g., sarcopenia) to evaluate its efficacy and further assess safety.
- Phase III Clinical Trials: Large-scale, multicenter trials involving hundreds or thousands of patients to confirm its effectiveness, monitor side effects, and compare it to commonly used treatments.
This entire process can take over a decade and cost hundreds of millions of dollars, with no guarantee of success. While the science is exciting, we are many years away from knowing if SLU-PP-332 will ever become a safe and effective medicine.

Studies / References
Below is a summary of key studies relevant to SLU-PP-332 and related compounds. Note the clear distinction between animal and human research.
- SLU-PP-332 and ERR Agonist Preclinical Evidence
– This peer-reviewed study demonstrates an ERRα/β/γ agonist enhances oxidative metabolism and endurance capacity in mice.
Source: Synthetic ERRα/β/γ agonist increases metabolic gene programs, mitochondrial function, and endurance in vivo.
https://pubmed.ncbi.nlm.nih.gov/36988910/ - GW501516 (PPARδ Agonist) Exercise-Mimetic Effects (Animal Study)
– Preclinical rodent research showing enhanced fatty acid oxidation and endurance with PPARδ activation.
Source: PPARδ agonism increases skeletal muscle fatty acid oxidation and alters metabolic parameters in mice.
https://pubmed.ncbi.nlm.nih.gov/14676330/ - GW501516 Enhances Endurance in Mice (Animal Study)
– Another rodent model showing increased running distance and endurance with GW501516.
Source: GW501516 enhances endurance exercise performance and alters metabolite profiles in mice.
https://pubmed.ncbi.nlm.nih.gov/25943561/ - Cancer-Related Findings with PPARδ Agonists (Rodent Model)
– Rodent research indicating potential tumor-promoting effects under long-term PPARδ activation, relevant for safety warnings.
Source: PPARδ activation enhances colitis-associated colorectal cancer progression in mice.
https://pubmed.ncbi.nlm.nih.gov/30391747/ - Urolithin A Human Trial – Mitochondrial & Endurance Outcomes
– Human clinical trial showing urolithin A improved mitochondrial gene expression and walking endurance in older adults.
Source: Randomized clinical trial of urolithin A supplementation on mitochondrial and muscle function in older adults.
https://pmc.ncbi.nlm.nih.gov/articles/PMC8777576/ - Urolithin A Human Study – Muscle Function in Older Adults
– Supports mechanistic relevance for mitochondrial health in humans.
Source: Urolithin A supplementation improves muscle function and biomarkers in older adults.
https://pmc.ncbi.nlm.nih.gov/articles/PMC9133463/ - NMN (Nicotinamide Mononucleotide) Human Study – Metabolic Effects
– Human clinical evidence that NMN improves insulin sensitivity and mitochondrial gene expression.
Source: Effects of NMN supplementation on metabolic health in postmenopausal women with prediabetes.
https://pubmed.ncbi.nlm.nih.gov/33888596/ - AICAR (Animal Research Example of Exercise Mimetics)
– Classic animal model demonstrating AMPK activation’s endurance-like effects. Useful for historical context.
Source: AMPK activator AICAR replicates metabolic effects of exercise in mice.
https://pubmed.ncbi.nlm.nih.gov/14676231/
Note: SLU‑PP‑332 is strictly an experimental research compound studied in animal models; it is not a supplement, performance enhancer, or DIY exercise shortcut, and attempting to obtain or use it outside a controlled laboratory setting is unsafe and illegal.
Conclusion: The Promise and Peril of the SLU-PP-332 Peptide
The SLU-PP-332 compound represents a fascinating frontier in aging science. As an experimental compound that activates the ERR/PGC-1α pathway, it offers a powerful demonstration of how a single molecule can mimic the profound metabolic and endurance benefits of exercise in animal models. Its potential as a future therapeutic for conditions marked by muscle wasting and mitochondrial dysfunction—such as sarcopenia or muscular dystrophy—is undeniably significant.
However, this promise must be tempered with extreme caution. The history of similar compounds, like Cardarine, provides a sobering reminder of the potential dangers of manipulating master metabolic switches. The complete absence of human safety and efficacy data means that SLU-PP-332 remains firmly in the realm of laboratory research. It is not a supplement, a performance enhancer, or a shortcut to health. It is an unanswered scientific question, and for now, the safest and most effective way to boost mitochondrial function and reap the rewards of endurance signaling remains the one we’ve known all along: consistent physical exercise.