Semaglutide has rapidly emerged as a cornerstone in the management of metabolic disease, marking a significant advancement in pharmacological interventions for type 2 diabetes and obesity. As a potent glucagon-like peptide-1 (GLP-1) receptor agonist, its influence extends far beyond simple glucose control, touching upon complex physiological pathways that govern appetite, cardiovascular health, and inflammation. This profound metabolic impact has sparked intense research into its broader applications, including its potential role in mitigating age-related conditions and protecting vital organs, such as the eyes. This article provides an evidence-based exploration of Semaglutide, detailing its mechanism of action, its established benefits in weight management, its complex relationship with eye health, and its place within the evolving landscape of metabolic therapies.
What is Semaglutide and How Does It Work?
Semaglutide is a synthetic analog of the human hormone GLP-1, which is naturally produced in the intestine in response to food intake. GLP-1 is an incretin hormone, meaning it enhances the secretion of insulin. However, natural GLP-1 has a very short half-life, being broken down by enzymes within minutes. Semaglutide was engineered with specific structural modifications that protect it from enzymatic degradation, extending its half-life to approximately one week and allowing for once-weekly administration.
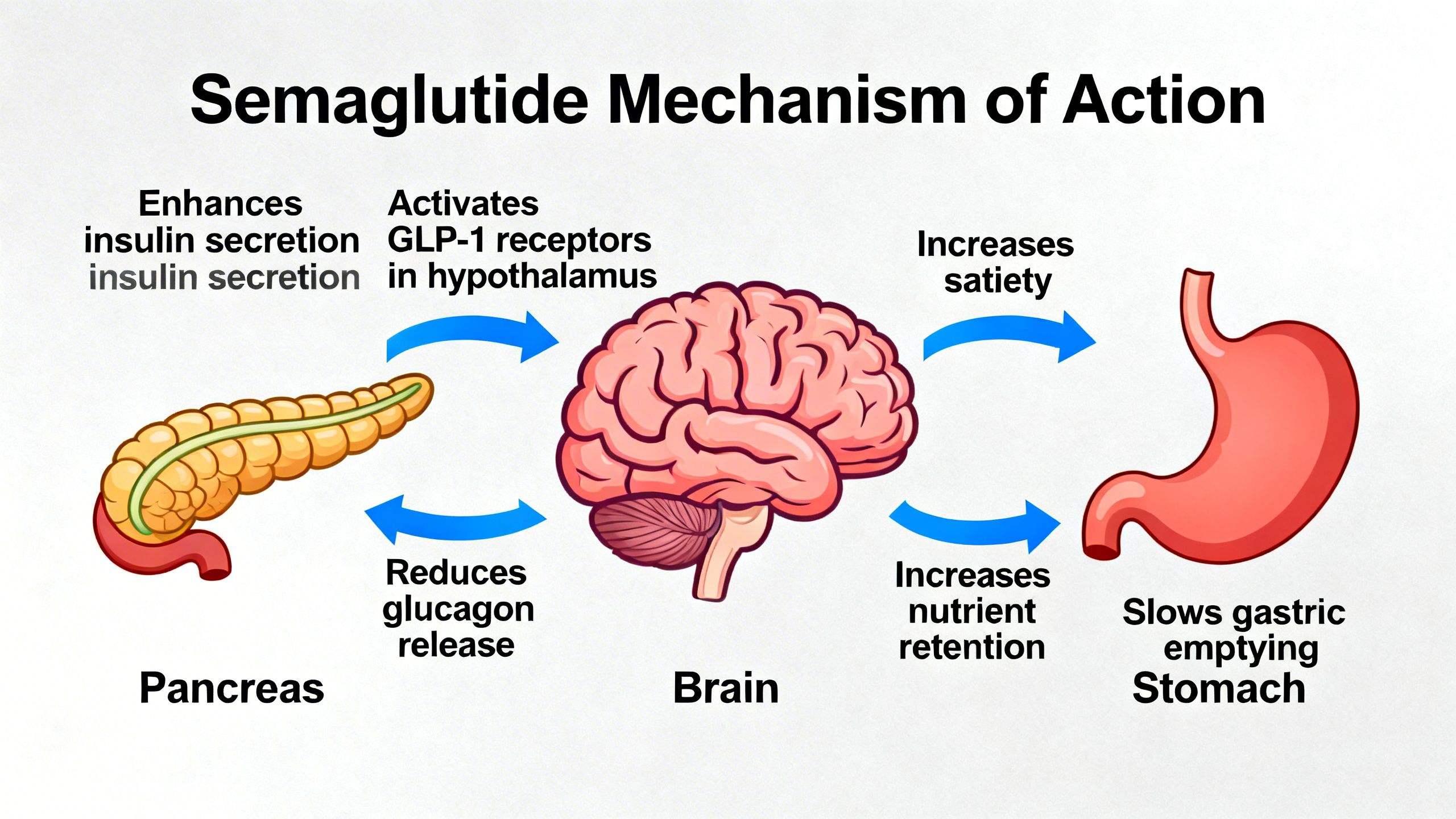
By binding to and activating GLP-1 receptors found throughout the body, Semaglutide initiates a cascade of metabolic improvements. Its therapeutic effects are not isolated to a single organ but are the result of a coordinated, multi-systemic response.
Mechanism of Action: A Multi-Pronged Approach
The question of how semaglutide works is best answered by examining its four primary physiological actions:
- Enhanced Insulin Secretion: Semaglutide stimulates the pancreas to release insulin in a glucose-dependent manner. This means it primarily acts when blood sugar levels are high, such as after a meal, and has a minimal effect when blood sugar is normal or low. This intelligent mechanism significantly reduces the risk of hypoglycemia compared to older diabetes medications.
- Glucagon Suppression: It acts on the alpha cells of the pancreas to decrease the secretion of glucagon, a hormone that signals the liver to produce and release glucose. By suppressing excessive glucagon, Semaglutide helps lower fasting and post-meal blood sugar levels, a key factor in addressing semaglutide and insulin resistance.
- Delayed Gastric Emptying: Semaglutide slows the rate at which food leaves the stomach. This delay contributes to a prolonged feeling of fullness and satiety after meals, naturally leading to a reduction in overall calorie intake.
- Central Nervous System Effects: GLP-1 receptors are also present in the brain, particularly in areas like the hypothalamus that regulate appetite and reward. Semaglutide crosses the blood-brain barrier to act on these receptors, reducing hunger, curbing food cravings, and decreasing the rewarding sensation associated with eating highly palatable foods.
Semaglutide for Weight Loss: Human Evidence
The most well-known of the semaglutide benefits outside of diabetes care is its profound effect on weight. The efficacy of semaglutide for weight loss has been robustly demonstrated in a series of large-scale human clinical trials known as the STEP (Semaglutide Treatment Effect in People with Obesity) program. These trials consistently showed that individuals with obesity or who were overweight with weight-related comorbidities achieved substantial and clinically meaningful weight loss.
In the landmark STEP 1 trial, participants without diabetes who received a weekly 2.4 mg dose of Semaglutide in conjunction with lifestyle intervention lost an average of 14.9% of their initial body weight over 68 weeks, compared to just 2.4% in the placebo group. A significant portion of participants, over one-third, achieved weight loss of 20% or more. These results led to its approval by regulatory agencies for chronic weight management, establishing it as one of the most effective pharmacological tools available for this purpose.
Beyond the Scale: Changes in Body Composition
While the total weight loss is impressive, it is crucial to consider the changes in body composition. The weight lost with Semaglutide is predominantly fat mass, which is highly beneficial for semaglutide metabolic health. However, a portion of the weight loss—typically around 20-40%—comes from lean mass, which includes muscle.
The loss of lean mass is a significant concern, especially in older adults who are already at risk of sarcopenia (age-related muscle loss). To mitigate this, it is strongly recommended that individuals using Semaglutide for weight management engage in regular resistance training and ensure adequate dietary protein intake. This strategy helps preserve muscle mass and function, ensuring that the benefits of fat loss are not compromised by a decline in physical strength and metabolic rate.
The Broader Metabolic Health Benefits of Semaglutide
The impact of Semaglutide extends well beyond the numbers on a scale. By addressing the root causes of metabolic dysfunction, it confers a wide range of health improvements.
Improving Insulin Sensitivity and Glycemic Control
As a primary treatment for type 2 diabetes, Semaglutide excels at improving glycemic control. It significantly lowers hemoglobin A1c (HbA1c), a key marker of long-term blood sugar levels. This effect is driven by its combined actions of boosting insulin secretion and suppressing liver glucose production. By reducing body weight and visceral fat—a major contributor to insulin resistance—Semaglutide helps the body’s cells become more responsive to insulin, improving overall glucose metabolism.
Cardiovascular Protection
Perhaps one of the most critical semaglutide benefits is its proven ability to protect the cardiovascular system. The SUSTAIN-6 and SELECT clinical trials provided definitive evidence of this effect. In patients with type 2 diabetes and high cardiovascular risk (SUSTAIN-6), Semaglutide reduced the risk of major adverse cardiovascular events (MACE), such as heart attack, stroke, and cardiovascular death, by 26%.
More recently, the SELECT trial studied individuals with pre-existing cardiovascular disease and obesity but without diabetes. The results were equally compelling, showing a 20% reduction in MACE for those treated with Semaglutide compared to placebo. These benefits are thought to be mediated through multiple pathways, including weight loss, improved blood pressure, better lipid profiles, and a direct reduction in semaglutide and inflammation within blood vessels.
Semaglutide and Inflammation
Chronic, low-grade inflammation is a key driver of many age-related diseases, from atherosclerosis to neurodegeneration. Obesity is a major source of this inflammation, as adipose (fat) tissue releases pro-inflammatory cytokines. By promoting significant fat loss, Semaglutide helps to quell this inflammatory state. Human studies have shown that treatment with Semaglutide leads to a significant reduction in systemic inflammatory markers like high-sensitivity C-reactive protein (hs-CRP). This anti-inflammatory effect is likely a major contributor to its cardiovascular benefits and may have protective implications for other organ systems, a key consideration for semaglutide aging research.
An Emerging Frontier: Semaglutide and Eye Health
The connection between systemic metabolic health and ocular disease is well-established. Conditions like diabetic retinopathy and age-related macular degeneration (AMD) are heavily influenced by factors like hyperglycemia, oxidative stress, and inflammation. This has led researchers to investigate the potential semaglutide effects on vision and overall eye health.
Diabetic Retinopathy: A Complex Relationship
Diabetic retinopathy is a leading cause of blindness caused by damage to the blood vessels of the retina. While long-term glycemic control is the best way to prevent its progression, the initial data on GLP-1 agonists presented a complex picture. Some early trials, including SUSTAIN-6, noted a paradoxical, short-term worsening of pre-existing diabetic retinopathy in a small subset of patients treated with Semaglutide.
It is crucial to understand the context of this finding. This phenomenon is not believed to be a direct toxic effect of the drug itself. Rather, it is thought to be a consequence of the rapid improvement in blood glucose control. Similar temporary worsening has been observed with other intensive glucose-lowering therapies, including insulin pumps. The prevailing hypothesis is that the rapid metabolic shift causes hemodynamic changes in the fragile retinal blood vessels, leading to transient worsening. Long-term data and meta-analyses suggest that over time, the profound benefits of improved metabolic health likely outweigh this initial risk, but it remains an important consideration for clinicians monitoring patients with pre-existing, advanced retinopathy.
Potential Protective Effects on the Retina and Vision
Beyond the complexities of diabetic retinopathy, there is growing interest in the potential protective role of semaglutide and eye health. Preclinical research has identified GLP-1 receptors in various retinal cells, suggesting a direct biological role in the eye. In animal models, GLP-1 agonists have demonstrated neuroprotective, anti-inflammatory, and anti-apoptotic (cell death-preventing) effects on the retina.
This has raised questions about a potential role for Semaglutide and AMD. Age-related macular degeneration is a leading cause of vision loss in older adults, driven by oxidative stress and inflammation in the macula. By systemically reducing inflammation and improving metabolic parameters, Semaglutide could theoretically create a less hostile environment for the aging retina. However, it must be emphasized that this is currently speculative. There are no large-scale human trials that have proven Semaglutide can prevent or treat AMD. This remains an active and promising area of research, but it is not an approved or established use for the compound.
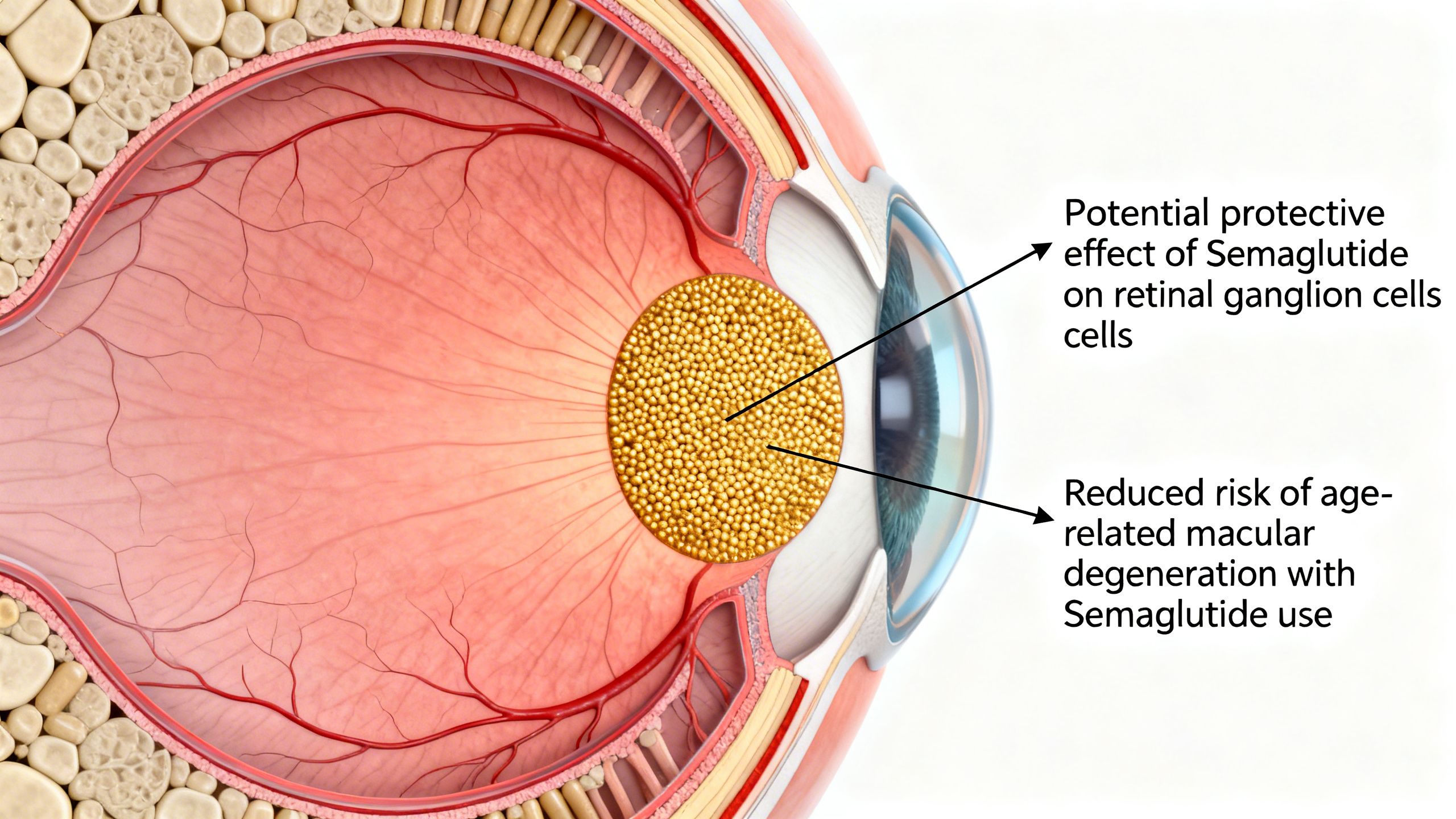
It is important to note that current research linking GLP-1 receptor agonists such as semaglutide to age-related macular degeneration is preliminary and observational, and does not establish causation or clinical benefit.
Semaglutide in the Context of Aging
The fundamental processes of aging are closely intertwined with metabolic health. Concepts like “inflammaging” (chronic, low-grade inflammation that accompanies aging) and immunosenescence are exacerbated by metabolic dysfunction. By targeting core pillars of metabolic disease—obesity, insulin resistance, and inflammation—Semaglutide may indirectly influence several aspects of the semaglutide aging trajectory.
Its benefits in reducing visceral fat, improving cardiovascular health, and lowering systemic inflammation address key drivers of age-related morbidity. Furthermore, research is exploring its effects on other age-related conditions. For example, by promoting weight loss, Semaglutide can significantly improve non-alcoholic fatty liver disease (NAFLD), a condition that can progress to cirrhosis. There is also preliminary but intriguing research into the potential neuroprotective effects of GLP-1 agonists in the context of Alzheimer’s and Parkinson’s disease, though this is still in the very early stages of investigation.
Safety, Side Effects, and Important Considerations
While Semaglutide offers powerful benefits, it is a potent medication with a distinct side effect profile and important safety considerations. A thorough understanding of the risks is essential for its appropriate use.
Common Side Effects
The most frequently reported semaglutide side effects are gastrointestinal in nature. These include:
- Nausea
- Vomiting
- Diarrhea
- Constipation
- Abdominal pain and bloating
These effects are typically most pronounced when starting the medication or after a dose increase. They are often transient and can be managed by starting at a low dose and titrating upwards very slowly over several weeks or months. Eating smaller, more frequent meals and avoiding high-fat foods can also help mitigate these symptoms.
Serious but Rare Risks
Beyond the common GI issues, there are several more serious, albeit rare, risks associated with Semaglutide:
- Thyroid C-Cell Tumors: Semaglutide carries a boxed warning from the FDA regarding thyroid C-cell tumors. This warning is based on studies in rodents where the drug caused an increase in these tumors. It is unknown if this risk translates to humans, as the C-cells in rodents are more sensitive. However, due to this potential risk, Semaglutide is contraindicated in patients with a personal or family history of medullary thyroid carcinoma (MTC) or in patients with Multiple Endocrine Neoplasia syndrome type 2 (MEN 2).
- Pancreatitis: Acute pancreatitis has been reported in patients taking GLP-1 receptor agonists. Patients should be advised of the hallmark symptoms (severe, persistent abdominal pain that may radiate to the back) and instructed to seek immediate medical attention if they occur.
- Gallbladder Disease: There is an increased risk of gallbladder-related issues, including gallstones (cholelithiasis) and gallbladder inflammation (cholecystitis), which may be related to rapid weight loss.
- Hypoglycemia: When used as a monotherapy, Semaglutide has a low risk of causing low blood sugar. However, this risk increases significantly when it is combined with other diabetes medications, particularly insulin or sulfonylureas.
The Evolving Landscape of Incretin Mimetics
Semaglutide is a leader in its class, but it is part of a rapidly advancing field of incretin-based therapies. Understanding its place in this landscape is key.
Semaglutide vs. Tirzepatide: The Dual Agonist
The most direct comparator and competitor to Semaglutide is Tirzepatide. While Semaglutide is a pure GLP-1 receptor agonist, Tirzepatide is a dual-agonist, activating both the GLP-1 and the GIP (glucose-dependent insulinotropic polypeptide) receptors. GIP is another incretin hormone that contributes to insulin release and appears to play a role in fat metabolism. Head-to-head clinical trials have shown that Tirzepatide can lead to even greater reductions in both HbA1c and body weight compared to Semaglutide, setting a new benchmark for efficacy. The discussion of semaglutide vs tirzepatide highlights the ongoing innovation in this space.
Other GLP-1 Receptor Agonists
Before Semaglutide, earlier GLP-1 receptor agonists like Liraglutide paved the way. Liraglutide requires a once-daily injection and is less potent for weight loss, but it established the class’s safety and efficacy. The development of Semaglutide represented a major step forward in terms of potency and convenience (once-weekly injection).
The Future: Triple Agonists and Combination Therapies
The next wave of innovation is already in late-stage development. Retatrutide, a triple-agonist for the GLP-1, GIP, and glucagon receptors, has shown unprecedented levels of weight loss in early trials, with average reductions exceeding 24% of body weight. Researchers are also exploring combination therapies, such as co-formulating a GLP-1 agonist with an amylin analog (another hormone involved in satiety) to achieve synergistic effects on weight and metabolism.
Studies / References
This section summarizes key human studies that form the evidence base for Semaglutide’s use.
- STEP 1 Trial (Once-Weekly Semaglutide in Adults with Overweight or Obesity): This 68-week randomized trial involved 1,961 adults with obesity but without diabetes. Participants receiving 2.4 mg of Semaglutide weekly achieved a mean weight loss of 14.9%, compared to 2.4% in the placebo group, both with lifestyle intervention. The main limitations were the high incidence of transient gastrointestinal side effects and a study population that was predominantly white and female.
Source: STEP 1 Trial Semaglutide Weight Loss (PubMed) - SUSTAIN-6 Trial (Semaglutide and Cardiovascular Outcomes in Patients with Type 2 Diabetes): This trial followed 3,297 patients with type 2 diabetes and high cardiovascular risk for approximately 2 years. Semaglutide was found to significantly reduce the risk of a composite cardiovascular outcome (death from cardiovascular causes, nonfatal heart attack, or nonfatal stroke) by 26% compared to placebo. A key limitation and point of discussion was the observation of an increased rate of diabetic retinopathy complications in the Semaglutide group, warranting further study.
- SELECT Trial (Semaglutide and Cardiovascular Outcomes in Obesity without Diabetes): This landmark trial enrolled 17,604 individuals with pre-existing cardiovascular disease and obesity but without diabetes. Over a median follow-up of nearly 40 months, weekly 2.4 mg Semaglutide reduced the risk of the primary MACE outcome by 20% versus placebo. This study definitively proved the cardiovascular benefits of Semaglutide extend to a non-diabetic population, though gastrointestinal side effects were a common reason for treatment discontinuation.
- GLP-1RAs Linked to Reduced Risk of AMD (Observational Cohort)
This retrospective cohort study found that long-term use of GLP-1 receptor agonists (including semaglutide) was associated with a lower hazard of developing both non-exudative and exudative forms of AMD compared with other medications like metformin and statins.
This large observational research suggests GLP-1RAs may be associated with reduced incidence of AMD compared with other drug classes — an important piece of evidence if you’re discussing potential retinal outcomes.
Conclusion: The Role of Semaglutide in Modern Medicine
Semaglutide represents a paradigm shift in the management of obesity and type 2 diabetes. Its powerful, multi-faceted mechanism of action delivers not only superior weight loss and glycemic control but also proven cardiovascular protection. By targeting the interconnected pathways of metabolism and inflammation, it has become an indispensable tool in modern medicine.
While its benefits are clear, its use requires a nuanced understanding of its side effect profile, particularly the common gastrointestinal issues and the rare but serious risks. Emerging research into its effects on other organ systems, including the potential for both benefits and risks related to semaglutide and eye health, underscores that we are still uncovering the full scope of its impact. As research continues and newer, even more potent incretin-based therapies arrive, Semaglutide will be remembered as the compound that truly revolutionized the pharmacological approach to metabolic disease and its many age-related consequences.
Frequently Asked Questions About Semaglutide
Why am I not losing weight on semaglutide?
Not losing weight on semaglutide is more common than many people expect, especially early on. Factors such as dose escalation speed, insulin resistance, sleep quality, diet composition, and metabolic adaptation can all influence results. Weight loss with semaglutide is typically gradual and varies significantly between individuals.
How long does it take for semaglutide to work?
Semaglutide usually begins affecting appetite and blood sugar regulation within the first few weeks, but visible weight loss may take longer. Many people report noticeable changes between 4 and 12 weeks as metabolic effects accumulate.
How long does it take for semaglutide to suppress appetite?
Appetite suppression often occurs before major weight changes. Some individuals feel reduced hunger within the first 1–2 weeks, while others require more time depending on metabolic health and sensitivity to GLP-1 receptor activation.
What do semaglutide before and after results typically show?
Semaglutide before and after comparisons often show gradual fat loss, reduced waist circumference, and improved metabolic markers rather than rapid scale changes. Results at 4 weeks are usually modest, with more meaningful changes appearing over several months.
Why does semaglutide sometimes stop working?
When semaglutide seems “not working,” it may be due to metabolic plateaus, changes in calorie intake, reduced protein consumption, or the body adapting over time. This does not necessarily mean the medication has failed, but that additional lifestyle or clinical adjustments may be needed.
Does semaglutide make you tired?
Fatigue is a reported side effect for some users, especially during early treatment phases. This may be related to reduced calorie intake, gastrointestinal effects, or temporary changes in blood sugar regulation.
How long do semaglutide side effects last?
Most semaglutide side effects—such as nausea, headache, or constipation—tend to improve within weeks as the body adapts. Persistent or worsening symptoms should always be discussed with a healthcare professional.
Does semaglutide need to be refrigerated?
Semaglutide storage requirements depend on formulation and manufacturer guidance. In general, unused medication is refrigerated, while in-use products may have specific room-temperature limits. Always follow pharmacy instructions.
Does semaglutide expire, and what happens if you use expired semaglutide?
Like all medications, semaglutide has an expiration date. Using expired semaglutide may reduce effectiveness and could increase safety risks, so expired products should not be used.
How long does semaglutide stay in your system?
Semaglutide has a long half-life and remains active in the body for several weeks after the last dose. This extended action contributes to its once-weekly dosing schedule.
What is the difference between semaglutide and tirzepatide?
The difference between semaglutide and tirzepatide lies in their mechanisms. Semaglutide targets the GLP-1 receptor, while tirzepatide activates both GLP-1 and GIP receptors, which may influence appetite control and metabolic outcomes differently.
Retatrutide vs semaglutide: how do they compare?
Retatrutide is an investigational multi-receptor agonist that targets GLP-1, GIP, and glucagon receptors. Compared to semaglutide, it is still under clinical evaluation and not yet widely available.
Liraglutide vs semaglutide: what’s the difference?
Liraglutide and semaglutide are both GLP-1 receptor agonists, but semaglutide has a longer duration of action, allowing for weekly dosing instead of daily injections.
Can semaglutide be used with vitamin B12?
Semaglutide with B12 is sometimes discussed due to concerns about fatigue or nutrient intake during weight loss. There is no universal requirement to combine them, and decisions should be individualized.
Is sublingual semaglutide or semaglutide drops effective?
Sublingual semaglutide and semaglutide drops are not FDA-approved formulations. Clinical evidence supporting their effectiveness and safety is limited compared to injectable forms.
Is compounded semaglutide the same as Wegovy?
Compounded semaglutide vs Wegovy comparisons often come up, but they are not identical. Wegovy is an FDA-approved product, while compounded versions vary in formulation, quality control, and regulatory oversight.
Where do people usually receive semaglutide treatment?
People often search for semaglutide clinics or weight loss clinics near them. Treatment should always be supervised by licensed medical professionals with experience in metabolic care.
Does semaglutide have implications beyond weight loss?
Beyond weight loss, semaglutide is being studied for its effects on insulin resistance, inflammation, cardiovascular risk, and potentially age-related conditions linked to metabolic dysfunction.
What does “semaglutide” mean and how is it pronounced?
Semaglutide is pronounced seh-MAG-loo-tide. It is a synthetic GLP-1 receptor agonist designed to mimic natural incretin hormones involved in appetite and glucose regulation.