In the rapidly advancing field of metabolic medicine, a new class of therapeutics is redefining the treatment landscape for obesity and related conditions. Building on the success of single and dual-agonist peptides, researchers have developed a novel compound with a three-pronged mechanism. This article provides an expert, evidence-based overview of Retatrutide (also known as LY-3437943), an investigational triple-agonist peptide showing unprecedented potential in early human trials. We will delve into its unique mechanism, review the clinical data, and explore its prospective role in addressing metabolic syndrome and promoting healthier aging.
Understanding the Incretin System and Beyond: The Foundation of Retatrutide
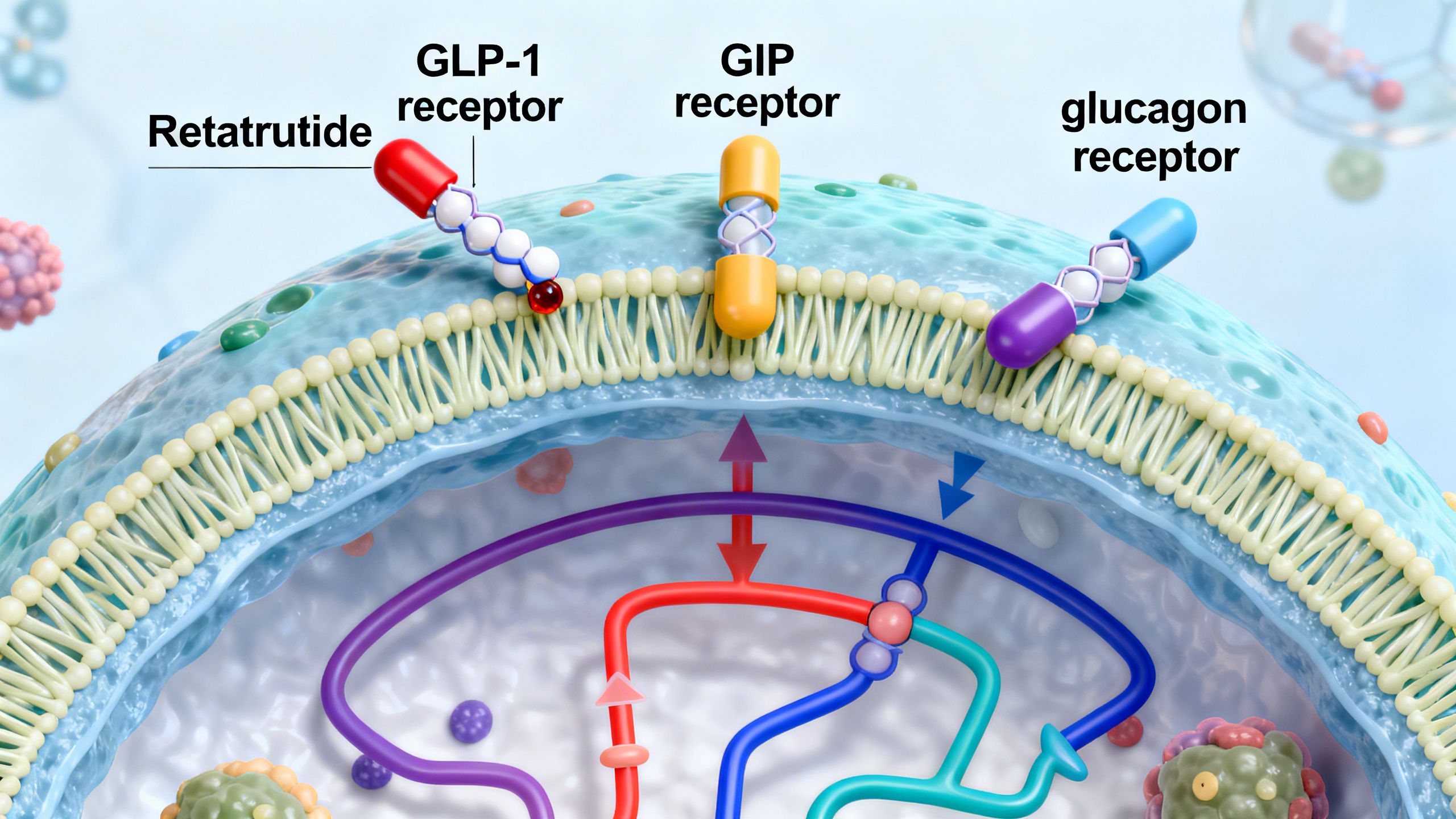
To appreciate the innovation of Retatrutide, it’s essential to understand the hormones it mimics. The human body uses a complex network of signals to regulate appetite, blood sugar, and energy balance. Retatrutide simultaneously activates three key receptors in this network: the GLP-1 receptor, the GIP receptor, and the glucagon receptor.
- Glucagon-Like Peptide-1 (GLP-1): Secreted by the gut after a meal, GLP-1 is a cornerstone of modern metabolic drugs. It enhances insulin secretion in response to glucose, slows stomach emptying to increase feelings of fullness, and acts directly on the brain to suppress appetite. Compounds like Semaglutide and Liraglutide are pure GLP-1 receptor agonists.
- Glucose-dependent Insulinotropic Polypeptide (GIP): Also an incretin hormone released from the gut, GIP was once thought to be less critical. However, research has shown it also stimulates insulin release and may play a role in how the body processes and stores fat. The dual-agonist Tirzepatide, which combines GIP and GLP-1 agonism, demonstrated superior efficacy to GLP-1 agonists alone, proving the value of this dual approach.
- Glucagon: This hormone, produced by the pancreas, is traditionally known for raising blood sugar levels by stimulating the liver to release glucose. While activating its receptor might seem counterintuitive for a diabetes or obesity drug, glucagon also has other crucial effects. It increases energy expenditure, promotes the breakdown of fat (lipolysis), and can help reduce fat accumulation in the liver. The challenge has always been to harness these benefits without causing hyperglycemia.
Retatrutide is the first compound to successfully combine the actions of all three into a single molecule. The hypothesis is that the powerful glucose-lowering and appetite-suppressing effects of GLP-1 and GIP agonism can offset the potential blood sugar-raising effect of glucagon, allowing its beneficial effects on energy expenditure and fat metabolism to shine through. This synergistic design represents a significant step forward in metabolic syndrome research.
The Mechanism of Action: How Does Retatrutide Work?
The power of the LY-3437943 peptide lies in its multi-faceted impact on the body’s metabolic machinery. By activating three distinct pathways simultaneously, it creates a powerful, synergistic effect that addresses obesity and metabolic dysfunction from multiple angles.
Synergistic Effects on Appetite and Satiety
A primary driver of weight gain is an imbalance in the hormones that control hunger and fullness. Retatrutide tackles this head-on. The GLP-1 component acts on appetite centers in the hypothalamus, sending strong signals of satiety. This reduces the desire to eat and helps control portion sizes.
The GIP component may further enhance these effects, potentially influencing the brain’s reward pathways associated with food. The combined action on both receptors, along with slowed gastric emptying, leads to a profound and sustained reduction in caloric intake that feels more manageable for individuals than willpower-based dieting alone.
Enhancing Glycemic Control
For individuals with insulin resistance or prediabetes, maintaining stable blood sugar is a constant challenge. Retatrutide offers robust support for glycemic control through the incretin effect. Both GLP-1 and GIP stimulate the pancreas to release insulin only when blood glucose is elevated, such as after a meal. This glucose-dependent mechanism is crucial because it significantly reduces the risk of hypoglycemia (dangerously low blood sugar), a common concern with older diabetes medications like sulfonylureas.
Furthermore, the glucagon receptor agonism is balanced by the potent insulin-stimulating effects of the other two components, creating a self-regulating system. This allows for effective blood sugar management without the sharp peaks and troughs that can damage blood vessels and organs over time.
Boosting Energy Expenditure and Fat Metabolism
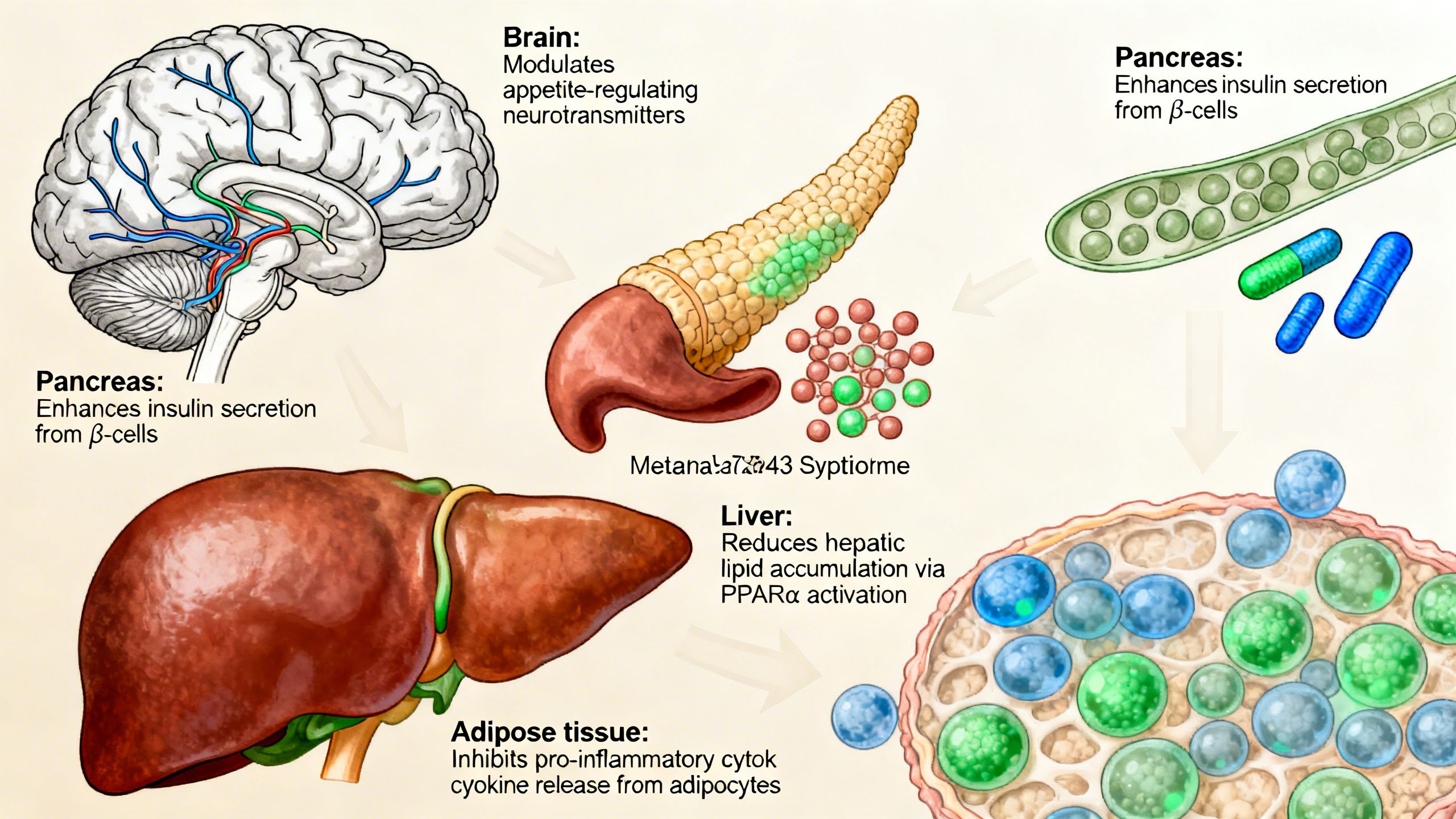
This is where Retatrutide truly distinguishes itself from previous generations of metabolic peptides. The inclusion of glucagon receptor agonism directly targets the body’s energy balance equation. It effectively ‘turns up the thermostat,’ increasing the basal metabolic rate and overall energy expenditure.
This mechanism has a particularly profound effect on the liver. Glucagon signaling helps to reduce hepatic steatosis (fatty liver) by promoting the breakdown and export of stored fats. It also stimulates lipolysis in adipose tissue, releasing stored fatty acids to be used for energy. This dual action not only drives weight loss but specifically targets the harmful visceral and ectopic fat deposits (like liver fat) that are most strongly linked to metabolic disease and systemic inflammation.
Human Evidence for Retatrutide: A Review of Clinical Trials
While the mechanism is compelling, the true test of any compound is its performance in human clinical trials. Retatrutide has produced some of the most striking results seen to date in the field of obesity medicine. It is important to note that Retatrutide is still an investigational drug and is not yet approved by the FDA or other regulatory agencies.
Landmark Phase 2 Trial in Obesity
The primary evidence for Retatrutide comes from a high-profile Phase 2 randomized, double-blind, placebo-controlled trial published in the New England Journal of Medicine in 2023. The study enrolled 338 adults with a BMI of 30 or higher (or 27 or higher with a weight-related comorbidity), but without type 2 diabetes.
The results were remarkable:
- Unprecedented Weight Loss: After 48 weeks of treatment, participants receiving the highest dose (12 mg weekly) achieved a mean weight reduction of 24.2% of their initial body weight. This translates to an average loss of nearly 58 pounds (26.4 kg).
- Dose-Dependent Efficacy: The weight loss effects were clearly dose-dependent, with lower doses producing more modest, yet still significant, results.
- Continued Weight Loss: Notably, the weight loss trajectory for the highest dose groups had not yet plateaued at the end of the 48-week study, suggesting that longer treatment duration could lead to even greater weight reduction.
- Cardiometabolic Improvements: Beyond weight loss, participants saw significant, dose-dependent improvements in key health markers. These included reductions in systolic and diastolic blood pressure, fasting glucose, insulin, and triglycerides, as well as an increase in beneficial HDL cholesterol.
- Normalization of Prediabetes: At the start of the trial, a significant portion of participants had prediabetes. By the end of the study, every single participant in the 8 mg and 12 mg dose groups who had prediabetes at baseline had reverted to normal glycemic status.
Effects on Non-alcoholic Fatty Liver Disease (NAFLD)
A particularly exciting finding from a sub-study of the Phase 2 trial was Retatrutide’s powerful effect on liver fat. NAFLD is extremely common in individuals with obesity and can progress to more severe liver inflammation (NASH) and cirrhosis. Using MRI scans to quantify liver fat, researchers found:
- At baseline, participants had an average liver fat content of around 20%.
- After 48 weeks, those on the 12 mg dose of Retatrutide saw their liver fat content decrease by an average of over 86%.
- Remarkably, nearly 86% of participants in the highest dose group achieved complete resolution of excess liver fat, defined as having a liver fat content below 5%.
These findings strongly support the proposed mechanism of glucagon receptor agonism and position Retatrutide as a leading candidate for the treatment of NAFLD.
Ongoing and Future Research (Phase 3 TRIUMPH Program)
Based on these highly successful Phase 2 results, Eli Lilly has launched an extensive Phase 3 clinical trial program named TRIUMPH. These larger, longer-term studies will evaluate the efficacy and safety of Retatrutide in diverse patient populations and for different indications, including:
- Chronic weight management in people with and without type 2 diabetes.
- Obstructive sleep apnea.
- Knee osteoarthritis in the context of obesity.
These trials will provide the definitive data needed for potential regulatory approval and will further clarify the long-term benefits and risks of this triple-agonist peptide.
Safety Profile and Side Effects of Retatrutide
No effective medication is without potential side effects, and it is crucial to approach Retatrutide with medical caution. The safety profile observed in the Phase 2 trial was consistent with the known effects of the incretin mimetic drug class.
The most commonly reported side effects were gastrointestinal in nature and included:
- Nausea
- Diarrhea
- Vomiting
- Constipation
These side effects were most common during the initial dose-escalation phase of the trial and tended to be mild to moderate in severity. Most participants were able to continue the medication as their bodies adapted. This highlights the importance of a slow and gradual dose titration schedule to improve tolerability.
As with all GLP-1 receptor agonists, there is a class-wide warning regarding a potential risk of thyroid C-cell tumors, based on findings in rodents. While this effect has not been established in humans, these drugs are contraindicated in patients with a personal or family history of medullary thyroid carcinoma or Multiple Endocrine Neoplasia syndrome type 2 (MEN 2). Other serious but rare risks associated with the class include acute pancreatitis and gallbladder disease. The long-term safety of Retatrutide is still being evaluated in the ongoing Phase 3 trials.
Crucially, Retatrutide is an investigational compound for research purposes only and is not approved for public use. It should not be used outside of a sanctioned clinical trial.
Retatrutide in Context: Comparison with Other Metabolic Peptides
The development of metabolic peptides has been a story of steady evolution. Understanding where Retatrutide fits requires comparing it to its predecessors and contemporaries.
Retatrutide vs. Tirzepatide (Dual GIP/GLP-1 Agonist)
Tirzepatide was the first dual-agonist to be approved, and it set a new standard for weight loss, with its SURMOUNT-1 trial showing approximately 21% weight loss at 72 weeks. Retatrutide, with its 24.2% weight loss at just 48 weeks in Phase 2, appears to be even more potent. The key difference is the addition of the glucagon agonist component in Retatrutide, which likely drives the enhanced effect on energy expenditure and liver fat reduction.
Retatrutide vs. Semaglutide (GLP-1 Agonist)
Semaglutide, a pure GLP-1 agonist, demonstrated around 15% weight loss in its pivotal STEP 1 trial. The superiority of both Tirzepatide and Retatrutide over Semaglutide suggests that activating additional metabolic pathways (GIP and glucagon) leads to a more comprehensive and powerful therapeutic effect. The triple-agonist mechanism of Retatrutide appears to offer the most robust impact on both sides of the energy balance equation: reducing energy intake and increasing energy expenditure.
Comparison with Growth Hormone Secretagogues (Tesamorelin, MK-677)
It is important to distinguish incretin-based peptides from another class of compounds that affect body composition: growth hormone secretagogues (GHS). Compounds like Tesamorelin (an approved drug for HIV-associated lipodystrophy) and MK-677 (a research chemical) work by stimulating the body’s own production of growth hormone.
This leads to an increase in lean muscle mass and a reduction in visceral fat. However, their mechanism is entirely different from Retatrutide. GHSs do not primarily target appetite or glycemic control in the same way. While both classes can lead to favorable changes in body composition, they are not interchangeable. Incretin mimetics are designed as comprehensive metabolic therapies, while GHSs have a more specific role related to the GH/IGF-1 axis.
Potential Applications in Aging and Degenerative Conditions
The implications of a compound like Retatrutide extend beyond simple weight loss and into the core drivers of age-related disease. Metabolic health is intrinsically linked to the aging process, and by addressing its root causes, Retatrutide may have a significant impact on healthspan.
- Combating ‘Metabolic Aging’: Metabolic syndrome—a cluster of conditions including high blood pressure, high blood sugar, excess body fat around the waist, and abnormal cholesterol levels—is a powerful accelerator of biological aging. By comprehensively targeting every component of this syndrome, Retatrutide could potentially slow the progression of age-related decline.
- Cardiovascular Health: The improvements in weight, blood pressure, lipids, and inflammation seen with Retatrutide directly reduce the major risk factors for heart attack and stroke, the leading causes of mortality in older adults.
- Neurodegeneration: There is emerging research, primarily from preclinical and early human studies with GLP-1 agonists, suggesting these peptides may have neuroprotective effects. Receptors for GLP-1 are found in the brain, and activating them may reduce inflammation and improve cellular function. While highly speculative for Retatrutide at this stage, it is an active area of investigation for the entire drug class in conditions like Alzheimer’s and Parkinson’s disease.
- Sarcopenic Obesity: A major concern with significant weight loss, especially in older adults, is the concurrent loss of muscle mass (sarcopenia). This can lead to frailty and reduced physical function. The unique metabolic profile of Retatrutide, particularly the role of glucagon, raises questions about its effect on muscle preservation. This will be a critical area to watch in the Phase 3 trial data, as preserving lean mass while aggressively targeting fat mass is the ultimate goal for healthy aging.
Studies / References
This section summarizes key human studies relevant to Retatrutide and its comparator compounds. These summaries are for informational purposes and are based on published, peer-reviewed data.
1. Triple–Hormone-Receptor Agonist Retatrutide for Obesity — A Phase 2 Trial (Jastreboff, A. M., et al., 2023, NEJM)
- Population: 338 adults with obesity (BMI ≥30 or ≥27 with a weight-related condition), without type 2 diabetes.
- Duration: 48 weeks.
- Intervention: Weekly subcutaneous injections of Retatrutide at various doses (1, 2, 4, 8, 12 mg) or placebo.
- Key Findings: The 12-mg dose group achieved a mean weight reduction of 24.2%. All participants with baseline prediabetes in the 8-mg and 12-mg groups achieved normoglycemia. Significant improvements in blood pressure, lipids, and insulin sensitivity were observed.
- Limitations: This was a Phase 2 trial with a limited duration and population size. The long-term efficacy and safety profile beyond 48 weeks are still under investigation in larger Phase 3 trials.
2. Retatrutide for Non-alcoholic Fatty Liver Disease (NAFLD) – Sub-study Analysis (Loomba, R., et al., presented at EASD 2023)
- Population: A subset of participants from the main Phase 2 obesity trial who had NAFLD, as assessed by MRI-Proton Density Fat Fraction (MRI-PDFF).
- Duration: 48 weeks.
- Intervention: Weekly subcutaneous Retatrutide vs. placebo.
- Key Findings: Retatrutide led to a profound, dose-dependent reduction in liver fat. At the 12-mg dose, 86% of participants achieved resolution of excess liver fat (defined as <5% liver fat content by MRI-PDFF).
- Limitations: This was a pre-specified exploratory analysis of a larger trial. The study was not powered to assess long-term liver outcomes like fibrosis progression or cirrhosis.
3. Tirzepatide Once Weekly for the Treatment of Obesity (Jastreboff, A. M., et al., 2022, NEJM)
- Population: 2,539 adults with obesity (BMI ≥30 or ≥27 with a weight-related condition), without type 2 diabetes.
- Duration: 72 weeks.
- Intervention: Weekly subcutaneous Tirzepatide (5, 10, or 15 mg) or placebo.
- Key Findings: The highest dose (15 mg) group achieved a mean weight reduction of 20.9% from baseline. The trial also demonstrated significant improvements across all measured cardiometabolic risk factors.
- Limitations: As a dual-agonist, this study cannot inform on the specific additional benefits or risks conferred by the glucagon-agonist component present in Retatrutide.
4. Once-Weekly Semaglutide in Adults with Overweight or Obesity (Wilding, J. P. H., et al., 2021, NEJM)
- Population: 1,961 adults with a BMI of 30 or greater (or 27 or greater with at least one weight-related comorbidity), without diabetes.
- Duration: 68 weeks.
- Intervention: Weekly subcutaneous Semaglutide (2.4 mg) or placebo, combined with a lifestyle intervention.
- Key Findings: The Semaglutide group achieved a mean weight loss of 14.9%, compared to 2.4% in the placebo group. Cardiometabolic risk factors also improved significantly.
- Limitations: This study evaluates a pure GLP-1 agonist, which has a less complex mechanism than dual or triple agonists and has shown lower peak efficacy for weight loss in cross-trial comparisons.
Conclusion: The Future of Metabolic Medicine
Retatrutide represents the current apex of incretin-based metabolic therapy. The early clinical evidence suggests it is a profoundly effective agent for inducing weight loss and normalizing key markers of metabolic health. Its unique triple-agonist mechanism—harnessing GLP-1, GIP, and glucagon—allows it to reduce appetite, improve glycemic control, and, crucially, increase energy expenditure, setting it apart from all predecessors.
The remarkable results in reducing liver fat underscore its potential as a transformative therapy for NAFLD, a condition with few effective treatments. As a focus of ongoing metabolic syndrome research, Retatrutide holds immense promise not just for weight management, but for addressing the interconnected web of diseases that drive premature aging.
While we must await the results of the comprehensive Phase 3 TRIUMPH program for definitive answers on long-term safety and efficacy, Retatrutide has opened a new chapter in our ability to pharmacologically manage obesity and its devastating consequences. It stands as a testament to the power of multi-targeted approaches in treating complex, systemic diseases.