In the ongoing battle against obesity and its associated metabolic complications, the scientific community is constantly searching for more effective and sustainable interventions. While lifestyle modifications remain the cornerstone of weight management, pharmacological agents are playing an increasingly vital role. Emerging from the pipeline of clinical research is the cagrilintide peptide, a novel compound that represents a significant advancement in our understanding and treatment of metabolic disease, with profound implications for healthy aging.
Cagrilintide is an investigational, long-acting amylin analog designed for weight management. Unlike the more widely known GLP-1 receptor agonists, cagrilintide works through a distinct but complementary pathway, primarily by mimicking the effects of the natural hormone amylin to induce a powerful sense of satiety. This unique mechanism of action, especially when combined with other metabolic drugs, positions cagrilintide as a potential game-changer for achieving substantial and lasting weight loss, thereby mitigating many of the downstream consequences of obesity that accelerate the aging process.
Understanding Amylin and Its Role in Metabolism
To appreciate the innovation of cagrilintide, one must first understand the hormone it emulates: amylin. Amylin is a peptide hormone that is co-secreted with insulin from the pancreatic beta cells in response to food intake. It is a key player in the intricate system of glucose homeostasis and energy balance, acting as a natural partner to insulin.
The primary functions of amylin in the body include:
- Promoting Satiety: Amylin acts on specific areas of the brain, most notably the area postrema, to signal a feeling of fullness, which helps to terminate meals and reduce overall calorie consumption.
- Slowing Gastric Emptying: It slows down the rate at which food moves from the stomach to the small intestine. This delay contributes to a prolonged sense of fullness and blunts the sharp post-meal spike in blood glucose.
- Suppressing Glucagon Secretion: Amylin inhibits the release of glucagon, a hormone that raises blood sugar levels, particularly after meals when blood glucose is already rising. This action helps maintain tighter glycemic control.
Despite these beneficial effects, native human amylin is not a viable therapeutic agent. It has a very short half-life, meaning it is cleared from the body rapidly, and it has a propensity to form amyloid fibrils, which can be problematic. This led researchers to develop amylin analogs—synthetic versions designed for greater stability and a longer duration of action. The first-generation amylin analog, pramlintide, was approved for use in patients with diabetes but requires multiple daily injections. Cagrilintide represents the next generation, engineered for once-weekly administration and greater efficacy.
The Mechanism of Action of the Cagrilintide Peptide
The cagrilintide peptide exerts its effects by selectively binding to and activating amylin receptors. Its design as a long-acting analog allows it to provide sustained signaling for appetite control throughout the week, a significant improvement over the body’s natural, short-lived amylin pulse.
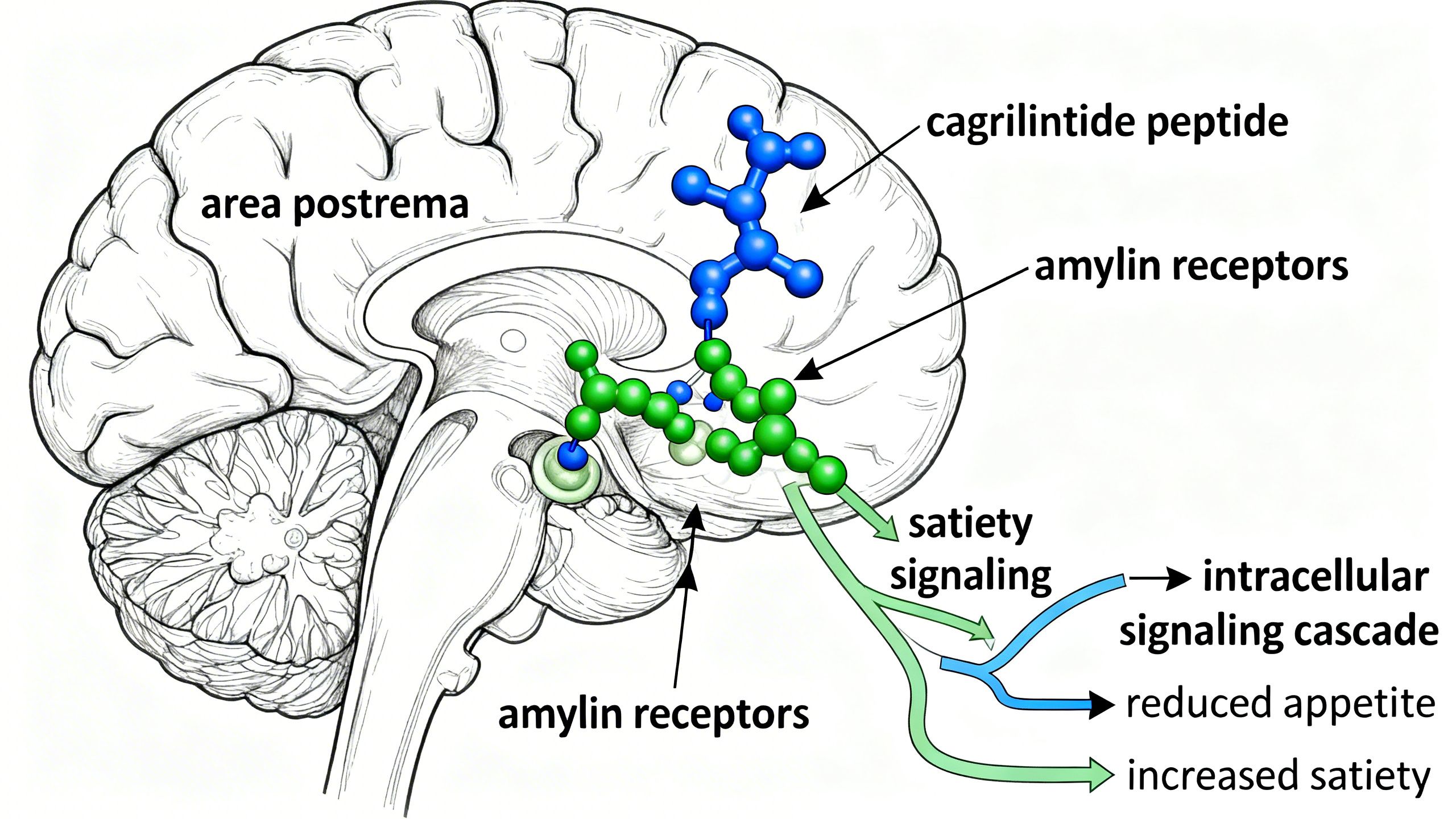
The core of the cagrilintide peptide mechanism of action is its potent effect on the central nervous system. By targeting amylin receptors in the hindbrain, it enhances the physiological signals of satiety that tell the body it is full. This leads to a reduction in hunger and a decreased desire to eat, resulting in lower overall energy intake. This is the primary driver of the Cagrilintide appetite control that has been observed in clinical studies.
Furthermore, cagrilintide’s ability to delay gastric emptying complements its central effects. By holding food in the stomach for a longer period, it not only helps control post-meal blood sugar but also provides a sustained feeling of fullness that can last for hours after eating. This dual action—central appetite suppression and peripheral slowing of digestion—creates a powerful and comprehensive approach to reducing calorie consumption without the feeling of deprivation often associated with traditional dieting.
Its long-acting profile is achieved through a process called acylation, where a fatty acid side chain is attached to the peptide molecule. This modification allows cagrilintide to bind to albumin, a common protein in the bloodstream, effectively creating a circulating reservoir of the drug that is released slowly over time. This clever biochemical engineering is what enables convenient once-weekly subcutaneous injections.
Human Evidence: Cagrilintide Clinical Trials for Weight Management
The promise of any new compound rests on robust evidence from human clinical trials. The cagrilintide peptide has undergone several key studies that have demonstrated its efficacy and safety for weight management.
Phase I trials were primarily designed to assess the safety, tolerability, and pharmacokinetic profile of cagrilintide. These studies involved dose-escalation protocols, where participants received increasing doses to identify a safe and effective range. The results confirmed the compound’s long half-life, supporting once-weekly dosing, and identified the most common side effects, which were predominantly gastrointestinal in nature.
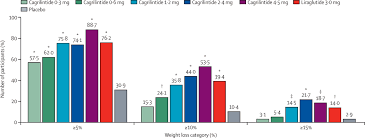
More revealing were the Phase II trials, which evaluated the Cagrilintide weight loss potential in larger populations. A landmark Phase II, multicenter, randomized, double-blind trial published in The Lancet investigated the effects of once-weekly cagrilintide in adults with overweight or obesity but without diabetes. Participants received various doses of cagrilintide (up to 4.5 mg) or a placebo for 26 weeks.
The results were compelling:
- Significant Weight Loss: Participants receiving the highest dose of cagrilintide (4.5 mg) achieved a mean body weight loss of 10.8% from baseline.
- Superiority to Placebo and Active Comparator: This was significantly greater than the 3.0% weight loss seen in the placebo group and the 9.0% loss in a group treated with liraglutide 3.0 mg, an established weight loss medication.
- Dose-Dependent Effect: The study showed a clear relationship between the dose of cagrilintide and the amount of weight lost, with higher doses leading to greater reductions.
- Improved Cardiometabolic Markers: Beyond weight loss, participants on cagrilintide saw improvements in waist circumference, lipid profiles, and other markers of metabolic health.
These findings established cagrilintide as a highly effective monotherapy for weight management, demonstrating a Cagrilintide fat loss effect that is clinically meaningful and competitive with existing treatments.
The Power of Combination: Cagrilintide with GLP-1 Agonists
While cagrilintide is potent on its own, perhaps its most exciting application is in combination with another class of powerful metabolic drugs: GLP-1 receptor agonists. This approach is based on the principle of multi-faceted therapy—targeting different biological pathways simultaneously to achieve a synergistic effect that is greater than the sum of its parts.
- Cagrilintide (Amylin Analog): Primarily reduces energy intake by promoting satiety.
- GLP-1 Agonists (e.g., Semaglutide): Reduce appetite, improve insulin secretion, and slow gastric emptying.
- Dual GIP/GLP-1 Agonists (e.g., Tirzepatide): Act on two incretin hormone receptors for even more profound effects on appetite, glucose control, and potentially energy expenditure.
Recognizing this potential, researchers began studying the co-administration of cagrilintide and semaglutide, a combination known as “CagriSema.” A Phase Ib trial investigated the safety and efficacy of this weekly combination in individuals with overweight. The results, presented at major medical conferences, were remarkable. The combination led to a mean weight loss of over 15% of body weight in just 20 weeks. This level of efficacy begins to approach that seen with bariatric surgery, marking a new pinnacle for pharmacological weight management.
The complementary mechanisms are key to this success. Cagrilintide provides a strong, sustained feeling of fullness, while semaglutide enhances this effect and adds robust benefits for glucose regulation. This dual-pronged attack on the drivers of obesity makes the combination exceptionally powerful.
Cagrilintide in Combination with Tirzepatide
Recent research and clinical discussions have highlighted the potential of combining cagrilintide with tirzepatide for enhanced weight management. Tirzepatide is a dual GIP and GLP-1 receptor agonist, and when paired with cagrilintide’s amylin-mediated satiety effects, this combination could target three distinct hormonal pathways that regulate appetite, glucose metabolism, and energy balance.
While formal clinical trials on the exact Cagrilintide dosage with tirzepatide are still emerging, preliminary studies suggest that dual therapy may produce greater weight loss and improved metabolic markers than either agent alone. This combination represents a promising frontier for individuals with overweight or obesity who need multi-faceted pharmacological support beyond lifestyle modifications.
Key Mechanistic Benefits:
- Enhanced satiety: Cagrilintide signals fullness via amylin receptors in the brain.
- Appetite suppression and glucose control: Tirzepatide activates GLP-1 and GIP pathways.
- Potential additive effects: Combining these mechanisms could reduce calorie intake and improve insulin sensitivity more effectively than monotherapy.

Cagrilintide and Retatrutide: Emerging Research Interest
Beyond Tirzepatide, the scientific community is exploring Cagrilintide + Retatrutide as a next-generation combination therapy. Retatrutide is an investigational triple agonist targeting GLP-1, GIP, and glucagon receptors, and early Phase 2 data indicate impressive weight-loss outcomes (up to ~28–30% in higher-dose cohorts).
The rationale for this combination lies in synergistic hormonal engagement:
- Cagrilintide: promotes fullness and slows gastric emptying.
- Retatrutide: simultaneously modulates GLP-1, GIP, and glucagon pathways, enhancing appetite suppression, glucose regulation, and potentially energy expenditure.
While no human trials have yet tested this dual approach, its mechanistic logic is compelling. It is important to emphasize that all use of investigational peptides outside clinical trials is unsafe. Current discussion is purely scientific, aiming to inform researchers, clinicians, and educated readers about potential future therapies.
Cagrilintide Peptide for Aging and Metabolic Health
The significance of the cagrilintide peptide extends far beyond the number on a scale; it directly intersects with the biology of aging. Obesity is not merely a cosmetic issue; it is a state of chronic, low-grade inflammation and metabolic dysfunction that dramatically accelerates the aging process and increases the risk of numerous age-related diseases.
By facilitating substantial and sustained weight loss, cagrilintide can help mitigate these risks. The reduction of excess adipose tissue, particularly visceral fat (the fat surrounding internal organs), is crucial. Visceral fat is a major source of inflammatory cytokines that contribute to “inflammaging”—the chronic inflammation that drives many diseases of aging.
Potential Cagrilintide peptide benefits for healthy aging include:
- Improved Cardiovascular Health: Significant weight loss is associated with lower blood pressure, improved cholesterol and triglyceride levels, and reduced strain on the heart. These changes can dramatically lower the lifetime risk of heart attack and stroke.
- Enhanced Glycemic Control: By improving the body’s sensitivity to insulin and promoting better blood sugar regulation, cagrilintide can help prevent or manage type 2 diabetes, a condition that severely impacts healthspan.
- Reduced Systemic Inflammation: The reduction of visceral fat through Cagrilintide fat loss directly lowers the burden of chronic inflammation, which is implicated in conditions ranging from arthritis to neurodegenerative diseases like Alzheimer’s.
- Potential for Lean Mass Preservation: A critical aspect of healthy aging is the preservation of muscle mass. Ideal weight loss interventions should primarily target fat mass while sparing lean tissue. Further Cagrilintide peptide research is needed to fully characterize its effects on body composition, but its mechanism focused on reducing energy intake may be more favorable for muscle preservation than severe caloric restriction alone.
Ultimately, by addressing a root cause of metabolic dysfunction—excess adiposity—cagrilintide offers a powerful tool to not only extend lifespan but, more importantly, to enhance healthspan, the period of life spent in good health.
Safety, Tolerability, and Dosage Considerations
No effective medication is without potential side effects, and cagrilintide is no exception. Based on human clinical trials, the safety profile of cagrilintide is generally considered acceptable, with most adverse events being predictable and manageable.
The most commonly reported side effects are gastrointestinal in nature and include:
- Nausea
- Vomiting
- Diarrhea
- Constipation
- Decreased appetite
These side effects are typically mild to moderate in severity, dose-dependent, and tend to be most pronounced when starting the medication or increasing the dose. They often diminish over time as the body adapts. To mitigate these effects, treatment is initiated at a low dose and gradually titrated upwards.
Regarding the Cagrilintide dosage, it is crucial to understand that cagrilintide is an investigational compound and is not approved for public use. The doses used in clinical trials, such as the once-weekly subcutaneous injection escalating to a target of 4.5 mg, are administered under strict medical supervision. Attempting to source or use research peptides like cagrilintide outside of a sanctioned clinical trial is extremely dangerous and carries significant health risks due to lack of quality control, unknown purity, and absence of medical oversight.

The Future of Cagrilintide Peptide Research
The journey of the cagrilintide peptide from laboratory to clinic is still underway. The promising Phase II results have paved the way for larger, more definitive Phase III trials. These trials will enroll thousands of participants and are designed to confirm the efficacy and long-term safety of cagrilintide, both as a monotherapy and in combination with other agents like semaglutide. The outcomes of these trials will be critical for seeking regulatory approval from bodies like the FDA and EMA.
Future Cagrilintide peptide research will likely explore its application in specific patient populations beyond general obesity. This could include individuals with obesity-related complications such as non-alcoholic steatohepatitis (NASH), heart failure with preserved ejection fraction (HFpEF), and polycystic ovary syndrome (PCOS). Researchers will also focus on long-term Cagrilintide peptide weight management, investigating its ability to not only induce weight loss but also to prevent the common problem of weight regain.
Studies / References
Below is a summary of key human studies that form the evidence base for cagrilintide. These summaries are for informational purposes and highlight the findings from controlled clinical research.
- Cagrilintide Phase 2 Dose-Finding Trial – multicentre, randomized, double-blind, placebo-controlled study showing significant dose-related weight loss with once-weekly cagrilintide vs placebo.
https://pubmed.ncbi.nlm.nih.gov/34798060/ - CagriSema (Cagrilintide + Semaglutide) Phase 3 Weight Loss Outcomes – combination therapy produced clinically meaningful and statistically significant weight loss (~20.4–22.7% at ~68 weeks).
https://pubmed.ncbi.nlm.nih.gov/40544433/ - CagriSema Combination Support (Semaglutide + Cagrilintide) in T2DM – shows improved HbA1c and weight outcomes vs monotherapy.
https://pubmed.ncbi.nlm.nih.gov/37364590/ - Phase 3 Clinical Data (REDEFINE Subanalysis) – confirms ~11.8% weight loss after 68 weeks with cagrilintide monotherapy in adults with obesity or overweight.
https://www.biospace.com/press-releases/novo-nordisk-presents-phase-3-data-for-next-generation-amylin-cagrilintide-leading-to-advancement-into-dedicated-clinical-programme/
Conclusion: Cagrilintide’s Place in Modern Medicine
The cagrilintide peptide stands at the forefront of a new wave of metabolic therapies that offer hope for profound and lasting health improvements. Its unique mechanism, centered on the powerful satiety signals of the amylin pathway, provides a distinct and complementary approach to weight management. While highly effective as a standalone agent, its true potential may be realized in combination with GLP-1 receptor agonists, where it has demonstrated the ability to produce weight loss that rivals surgical interventions.
By enabling significant Cagrilintide appetite control and subsequent fat loss, this investigational compound has the potential to do more than just treat obesity. It can help unwind the complex metabolic dysfunction that drives many diseases of aging, offering a pathway to a longer, healthier life. As Phase III trials progress, the medical community watches with anticipation. Cagrilintide may soon become a cornerstone of treatment, reshaping the future of Cagrilintide peptide weight management and metabolic medicine. However, it must be unequivocally stated that cagrilintide remains an unapproved, investigational drug and should not be used outside the confines of a properly regulated clinical trial.