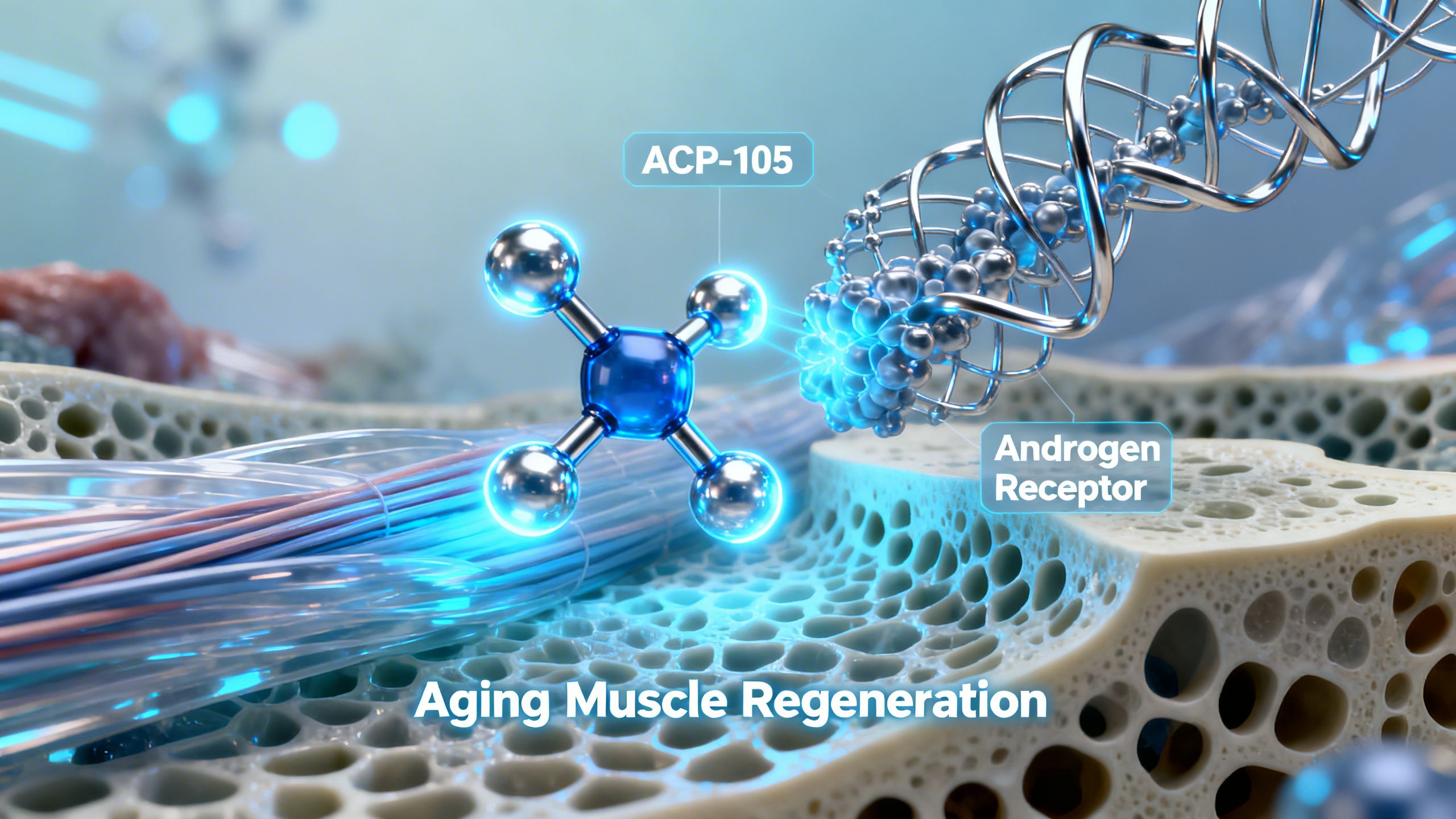
Selective Androgen Receptor Modulators, or SARMs, represent a class of investigational compounds designed to harness the therapeutic benefits of androgens with improved tissue selectivity. Among these is ACP-105, a non-steroidal SARM developed by Acadia Pharmaceuticals. While it has generated significant interest within research and biohacking communities, it’s crucial to approach the topic with a clear understanding of its scientific context. The primary goal behind ACP-105 was to create a compound that could provide the muscle- and bone-building effects of testosterone without the associated androgenic side effects, such as prostate enlargement or hair loss.
However, a critical distinction must be made from the outset: the vast majority of information regarding ACP-105 comes from pre-clinical animal studies. Human data is exceptionally scarce, making any discussion of its effects, benefits, or safety profile in humans largely speculative. This article will provide a comprehensive, evidence-based review of ACP-105, delving into its mechanism of action, the findings from pre-clinical research, its potential applications, and the significant safety considerations and unknowns that surround its use. We will navigate the science as it currently stands, separating established facts from extrapolation and hypothesis.
Understanding the Androgen Receptor and the SARM Concept
To appreciate how ACP-105 is designed to work, one must first understand its target: the androgen receptor (AR). Androgen receptors are proteins found inside cells throughout the human body. When activated by androgenic hormones like testosterone or dihydrotestosterone (DHT), they travel to the cell’s nucleus and influence gene expression, essentially telling the cell what to do.
This activation has different effects depending on the tissue:
- Anabolic Tissues: In muscle and bone, AR activation stimulates protein synthesis and increases bone mineral density. This leads to muscle growth (hypertrophy) and stronger bones.
- Androgenic Tissues: In tissues like the prostate, skin, and hair follicles, AR activation can lead to prostate growth, increased sebum production (acne), and male pattern baldness in genetically predisposed individuals.
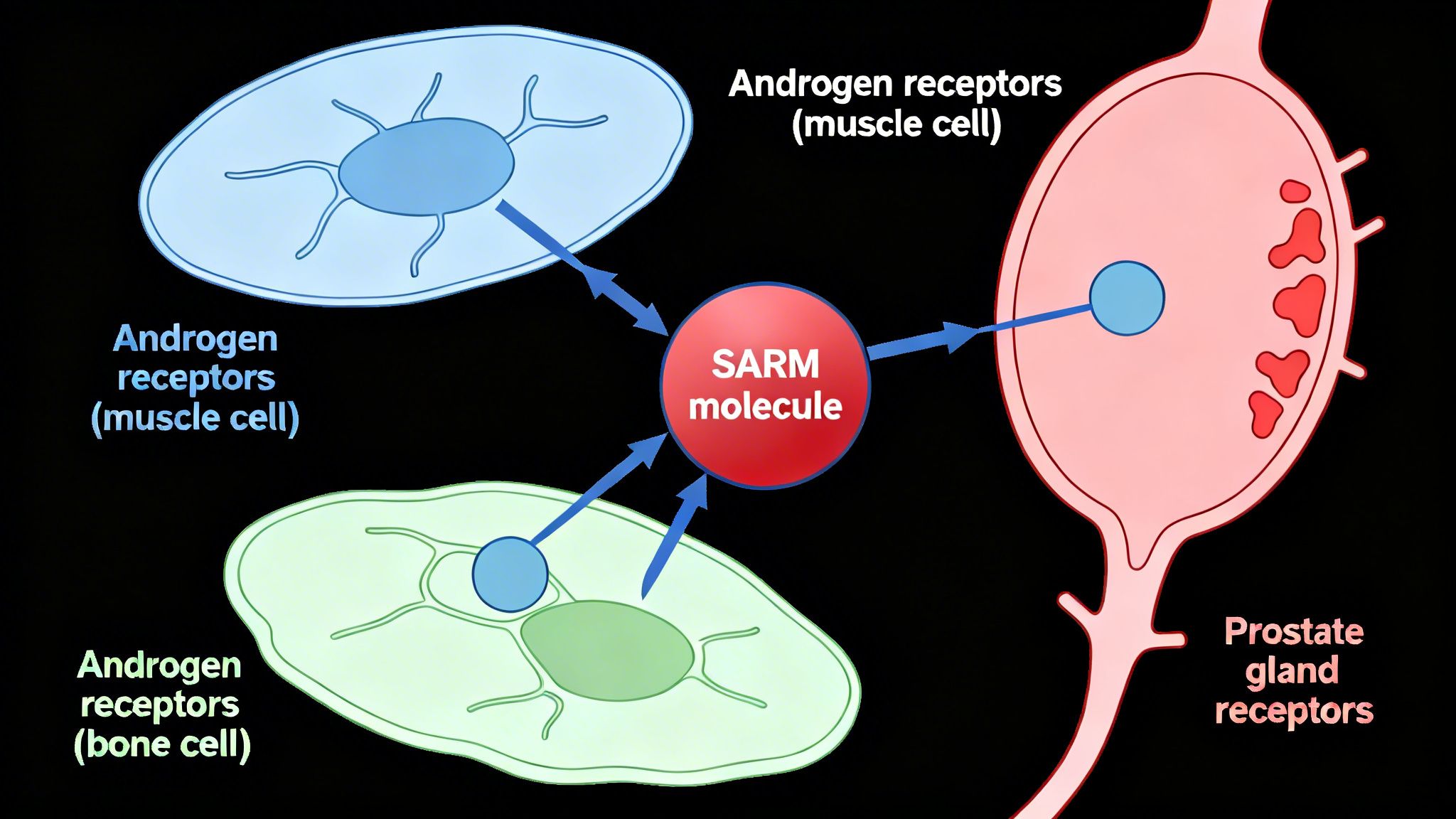
The therapeutic challenge with traditional anabolic-androgenic steroids (AAS) is that they activate ARs indiscriminately, leading to both desired anabolic effects and undesired androgenic side effects. The revolutionary concept behind SARMs is tissue selectivity. They are designed to bind to the AR and act as potent activators (agonists) in anabolic tissues like muscle and bone, while having a much weaker effect (or even acting as blockers, or antagonists) in androgenic tissues like the prostate. This theoretical selectivity is the cornerstone of their potential as safer therapeutic agents for conditions like age-related muscle wasting (sarcopenia), osteoporosis, and cachexia.
The Mechanism of Action of ACP-105
ACP-105 functions by binding directly to the androgen receptor. In laboratory (in-vitro) studies, it has demonstrated a high binding affinity for the AR, meaning it attaches to the receptor effectively. However, its key characteristic is that it appears to be a partial agonist. This is a crucial distinction from testosterone, which is a full agonist.
A full agonist, like testosterone, activates the receptor to its maximum capacity. A partial agonist, like ACP-105 is theorized to be, binds to the receptor but elicits a sub-maximal response. This ‘dimmer switch’ effect is a key part of its proposed safety profile. By not fully activating the receptor, it may provide sufficient anabolic signaling in muscle tissue to promote growth without reaching the threshold required to cause significant growth in the prostate or other androgenic tissues.
Pre-clinical data suggests that ACP-105’s anabolic potency is significant, though perhaps not as potent as some other SARMs in development. The goal isn’t necessarily to be the strongest compound, but to have the best ratio of anabolic-to-androgenic effects. This ratio, known as the therapeutic index, is the ultimate measure of a SARM’s success in achieving tissue selectivity.
Pre-Clinical Evidence: What Animal Studies Suggest
It is imperative to reiterate that animal studies provide foundational knowledge but do not guarantee the same results in humans. Metabolic differences, dosing, and disease models can all influence outcomes. The primary evidence for ACP-105’s potential comes from a key study involving irradiated mice.
This research was designed to model the side effects seen in cancer patients undergoing radiotherapy, which can include significant muscle wasting and loss of motor function. In this study, researchers compared the effects of ACP-105 to testosterone and a placebo.
The key findings were:
- Restored Motor Function: The mice treated with ACP-105 showed a significant improvement in motor performance, as measured by the rotarod test (an apparatus that tests coordination and endurance). This suggests a potential neuro-muscular benefit.
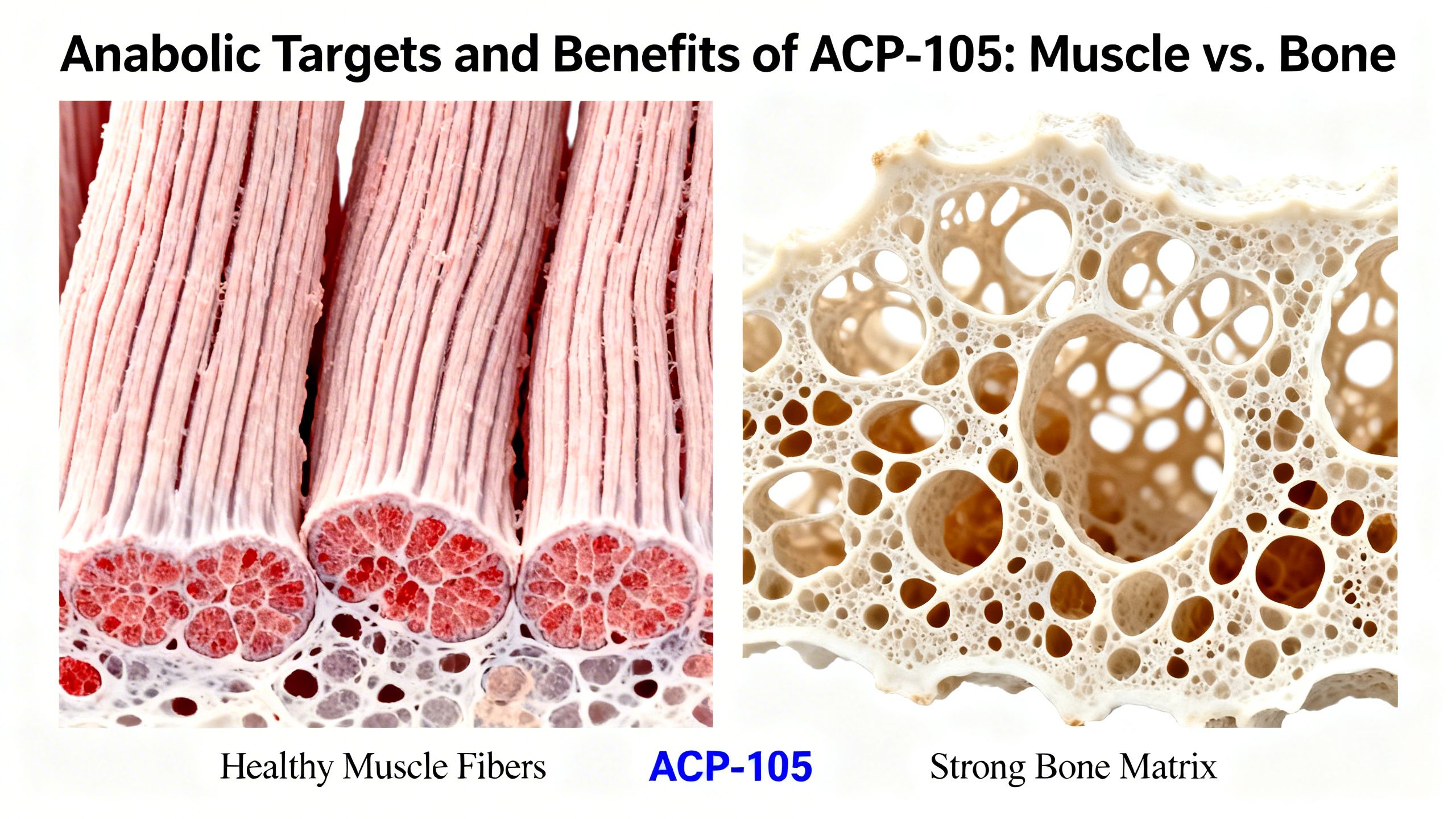
- Anabolic Activity: ACP-105 demonstrated clear anabolic effects on the levator ani muscle, a proxy used in rodent studies to measure muscle-building activity. Its potency was calculated to be approximately 66% that of testosterone in this regard.
- Prostate Sparing: This was the most significant finding. While producing two-thirds of the anabolic effect of testosterone, ACP-105 exerted only about 21% of the androgenic activity in the prostate. This demonstrated a favorable dissociation between anabolic and androgenic effects in this animal model.
These findings provided the proof-of-concept that ACP-105 could function as a true SARM, offering muscle-supportive benefits with a substantially lower risk of impacting the prostate. This study is the main reason ACP-105 is discussed as a potential therapeutic for muscle-wasting conditions. However, this is just one animal study, and the leap to human application is a large one that has not yet been successfully bridged in published literature.

The Scarcity of Human Research on ACP-105
This is the most critical section for anyone considering or researching this compound. Despite the promising animal data, there is a profound lack of published human research on ACP-105. Acadia Pharmaceuticals did initiate a Phase 1 clinical trial (NCT00393935) in the mid-2000s. A Phase 1 trial is the first step in human testing and is designed to evaluate a new drug’s safety, tolerability, and pharmacokinetic profile (how it’s absorbed, distributed, metabolized, and excreted) in a small group of healthy volunteers.
However, the full, peer-reviewed results of this trial have never been made public. The reasons for this are unknown—the compound may have shown a poor safety signal, lacked efficacy, or the company may have simply shifted its strategic priorities. Without this data, it is impossible to make any evidence-based conclusions about:
- The effective or safe dosage of ACP-105 in humans.
- Its true anabolic effects on human muscle mass or strength.
- The extent of its side effects, including testosterone suppression or impacts on cholesterol.
- Its long-term safety profile.
Any information found online regarding human dosages, cycle lengths, or specific outcomes is based on anecdotal reports from the grey market, not controlled scientific study. This makes personal use of the compound an N-of-1 experiment with significant unknown risks.
Potential Benefits Explored in Research (Hypothetical & Extrapolated)
Given the lack of human data, this section must be viewed as a discussion of potential applications based on the compound’s mechanism and pre-clinical results. These are not proven ACP-105 benefits in humans.
Muscle Growth and Strength (Anabolic Potential)
The primary interest in ACP-105 is for its potential to increase muscle mass and strength. By activating androgen receptors in skeletal muscle, it could theoretically stimulate muscle protein synthesis, leading to hypertrophy. This makes it a compound of interest for conditions characterized by muscle loss:
- Sarcopenia: The progressive loss of muscle mass and function with age. A selective, prostate-sparing anabolic agent could be highly beneficial for the elderly.
- Cachexia: Severe muscle wasting associated with chronic diseases like cancer, AIDS, or heart failure.
- Post-Surgical Recovery: Helping patients regain muscle mass and strength after prolonged bed rest or major surgery.
For the bodybuilding community, the interest in ACP-105 for muscle growth stems from this same mechanism, seeking anabolic effects without the harsh side effects of traditional steroids.
Bone Health
Androgen receptors are also present in bone tissue and play a vital role in maintaining bone mineral density. Testosterone deficiency is a known risk factor for osteoporosis in men. A SARM like ACP-105 could potentially stimulate ARs in bone, helping to increase bone formation and reduce resorption. This could make it a candidate for studying as a treatment for osteoporosis in both men and women, as it would not cause virilization (development of male characteristics) in female users.
Cognitive and Neurological Function
The finding that ACP-105 improved motor performance in mice is intriguing. Androgens are known to have effects on the central nervous system, influencing mood, libido, and cognitive function. The potential for a SARM to provide neuroprotective or performance-enhancing benefits without causing significant androgenic side effects is an area of active research, though it is highly speculative for ACP-105 specifically.
Safety Profile and Potential Side Effects of ACP-105
Discussing the safety of an unapproved compound with no published human trial data is challenging. The following information is extrapolated from the known side effects of the SARM class as a whole, which have been observed in clinical trials of other compounds like Ostarine and LGD-4033. These ACP-105 side effects are potential risks that any user would face.
Testosterone Suppression
This is arguably the most predictable and significant side effect. When the body detects an external androgenic compound activating the AR, it signals the brain to shut down its own production of testosterone. This is done through the Hypothalamic-Pituitary-Testicular Axis (HPTA).
- The hypothalamus reduces its release of Gonadotropin-Releasing Hormone (GnRH).
- This causes the pituitary gland to reduce its output of Luteinizing Hormone (LH) and Follicle-Stimulating Hormone (FSH).
- Without the LH signal, the testes stop producing testosterone.
All SARMs, including ACP-105, are known to cause dose-dependent testosterone suppression. The term ACP-105 suppression refers to this phenomenon. While some marketers claim certain SARMs are ‘non-suppressive’ at low doses, any dose sufficient to produce noticeable muscle growth will almost certainly cause some degree of HPTA shutdown. After a cycle, users often experience symptoms of low testosterone (fatigue, low libido, depression) until their natural production recovers, a process that can take weeks or months. This is why many recreational users employ a Post-Cycle Therapy (PCT) protocol, another non-medically-approved practice.
Impact on Lipid Profiles
Another common side effect observed in human trials of other SARMs is a negative impact on cholesterol levels. Specifically, SARMs tend to suppress High-Density Lipoprotein (HDL), often called ‘good’ cholesterol, which is responsible for removing excess cholesterol from the bloodstream. Some may also elevate Low-Density Lipoprotein (LDL), or ‘bad’ cholesterol. This combination can increase the long-term risk of atherosclerosis and cardiovascular disease. The extent to which ACP-105 affects human lipid profiles is completely unknown.
Liver Health (Hepatotoxicity)
Unlike many oral anabolic steroids that are ‘methylated’ and known to be harsh on the liver, most SARMs are non-methylated. They are generally considered to have a lower risk of causing direct liver damage. However, the risk is not zero. Any substance processed by the liver carries a potential for strain, and without human safety data, it is impossible to rule out hepatotoxicity, especially at high doses or in individuals with pre-existing liver conditions.
The Dangers of the Unregulated Market
This is a critical safety warning. Since ACP-105 is not an approved drug, anyone looking for ACP-105 for sale will find it only on the grey market, sold by ‘research chemical’ websites. These products are not subject to any regulatory oversight.
Multiple independent studies have analyzed the contents of products marketed as SARMs online, with alarming results:
- Wrong Compound: A significant percentage of products do not contain the SARM advertised on the label. Instead, they may contain a different, cheaper SARM, or even a prohormone or anabolic steroid.
- Incorrect Dosage: The actual dosage in the product can be much higher or lower than what is stated, making controlled administration impossible.
- Contamination: Products may be contaminated with other undeclared substances, including heavy metals or other toxic compounds.
Buying ACP-105 online is a significant gamble. There is no way to verify the identity, purity, or dosage of the substance without independent laboratory testing. This exposes the user to a host of unknown risks beyond those inherent to the compound itself.

Conclusion: The Current State and Future of ACP-105
In summary, ACP-105 is an investigational SARM that showed significant promise in early, pre-clinical animal research. Its ability to promote anabolic effects with substantially reduced androgenic activity in the prostate made it a compound of great theoretical interest for treating muscle-wasting conditions associated with aging, disease, and medical treatments. Its mechanism as a partial agonist further suggested a potentially favorable safety profile.
However, the promise of ACP-105 remains unrealized and unproven. The critical gap in the evidence is the near-complete absence of published human trial data. Without this information, any discussion of its efficacy, side effects, or proper dosing in humans is purely speculative and based on anecdotal reports from a dangerous, unregulated market. The known class-wide side effects of SARMs, including testosterone suppression and adverse lipid changes, are almost certain to apply to ACP-105, but to an unknown degree.
For now, ACP-105 must be regarded strictly as an experimental chemical, not a supplement or therapeutic agent. Its journey from a promising molecule in the lab to a proven medicine for patients has stalled, and until robust human clinical trials are conducted and published, its use remains an experiment with significant and poorly understood risks.